
Adult intracranial infection caused by an extended-spectrum-beta-lactamase-producing strain of hypervirulent Klebsiella pneumoniae: a case report
Introduction
Klebsiella pneumoniae, belonging to the gram-negative aerobic bacteria, is commonly found in communities and healthcare sites. Klebsiella pneumoniae is often parasitic in the human respiratory tract, intestinal tract, and biliary tract (1). It is an important conditional pathogen related to several infectious diseases, such as pneumonia, bacteremia, sepsis, meningitis, pyogenic liver abscess, and urinary tract infections. In some severe conditions, immunocompromised patients may even experience life-threatening diseases such as sepsis and purulent meningitis (2). Klebsiella pneumoniae represents an infrequent cause of meningitis in South-East Asia and North-East Asia, frequently associated with septic metastatic complications. And the in-hospital mortality rate of patients with Klebsiella pneumoniae meningitis is 8.0%, which increases substantially for those older than 45 years (3).
Along with the development of bacterial virulence and the changes in capsular polysaccharides, a new variant of Klebsiella pneumoniae has occurred, namely hypervirulent Klebsiella pneumoniae (hvKP). The hvKP has unique phenotypic and genotypic characteristics, for instance, high mucus phenotype, special serotype, and carrying special virulence genes (4). It can lead to a higher infectious rate in patients with low immune function, which also manifests with more severe clinical symptoms. It is traditionally considered that there is no overlap of the hvKP genome between high virulence and multi-drug resistance; most hvKPs are sensitive to commonly used antibiotics (5). Therefore, patients with hvKP infection require timely administration of antibiotics.
The most important mechanism of drug resistance in Klebsiella pneumoniae is to produce the extended-spectrum-beta-lactamases (ESBLs) (6). The ESBLs are β-lactamases that can hydrolyze penicillins, cephalosporins, and monocyclic amides, it was reported that the detection rate of ESBLs-producing Klebsiella pneumoniae was 27.4% in 2015 (7). With the wide application of new antibiotics such as carbapenems, the drug resistance of Enterobacteriaceae has increased, and carbapenem-resistant and fully drug-resistant Klebsiella pneumoniae have emerged (8). A study has identified that some phenotypes of Klebsiella pneumoniae can produce 1 or more β-lactamases; their plasmids often carry aminoglycosides, quinolones, and other antimicrobial resistance genes at the same time (9). Till now, the diagnosis and treatment of intracranial infection caused by an ESBLs-producing Klebsiella pneumoniae are remains difficult, corresponding guidelines on empiric treatment should be considered in the management of bacterial meningitis.
Here, we report a case of adult intracranial infection caused by ESBL-producing hvKP in a 65-year-old female, this case highlights to us that the effective and proper identification of these ESBL-producing Klebsiella pneumonia is a key step in the rational prevention and treatment for infectious patients. We present the following article in accordance with the CARE reporting checklist (available at https://atm.amegroups.com/article/view/10.21037/atm-22-3805/rc).
Case presentation
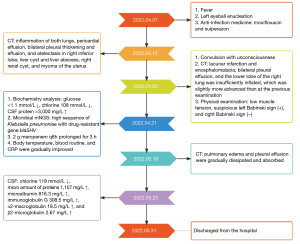
A 65-year-old female who had a chief complaint of recurrent fever for more than 1 month, nausea, and vomiting for 1 week was admitted to our hospital. She had no obvious induction of fever, and her highest body temperature was 38 ℃. No obvious clinical signs were found by physical examination. Routine blood test results showed the following: neutrophil count 6.96×109/L ↑, monocyte count 0.86×109/L ↑, lymphocyte percentage 14.9% ↓, red blood cell count 3.17×1012/L ↓, hemoglobin content 87 g/L ↓, hematocrit 0.28 L/L ↓, average red blood cell hemoglobin concentration 312 g/L ↓, platelet count 363×109/L ↑, platelet distribution width 8.3 fl ↓, and C-reactive protein (CRP) 52.01 mg/L ↑. Her blood routine test showed a gradually decreased platelet count, with abnormal infection indicators. Bone puncture showed megakaryocyte maturation disorder, and blood culture indicated Klebsiella pneumoniae sepsis.
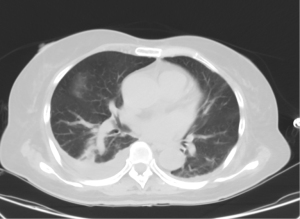
She was then admitted to the hematology department and diagnosed with “secondary infectious thrombocytopenia, gram-negative bacilli septicemia (Klebsiella pneumoniae), liver abscess, bilateral lung inflammation, type 2 diabetes, and hypertension grade 3 (extremely high risk)”. She was given vancomycin, caspofungin, dexamethasone, and posaconazole oral suspension for therapy. Liver abscess puncture and drainage treatment were performed with the assistance of the interventional oncology department. The inflammatory indexes tested by laboratory tests decreased significantly after treatment. During this period, light perception disappeared in the left eye, accompanied by eyelid redness and pain, purulent secretion, repeated fever, and left-sided headache. The diagnosis made by the ophthalmology department was as follows: endogenous endophthalmitis (left), orbital cellulitis (left), rubeosis iridis (left), exudative retinal detachment (left), and diabetic retinopathy (right). Intravitreal injection with vancomycin and ceftazidime was given 3 times into the left eye, the symptoms were relieved, and the patient’s current systemic infection was under control. On 7 April, 2022, the patient was treated with left eyeball enucleation, and she had a fever again after the operation. After the operation, anti-infection medicine including moxifloxacin and sulperazon was administered; however, her temperature elevated again. The computed tomography (CT) examination images performed on 12 April, revealed inflammation of both lungs, pericardial effusion, bilateral pleural thickening and effusion, and atelectasis in right inferior lobe (Figure 1); liver cyst and liver abscess, right renal cyst, and myoma of the uterus.
She had a history of hypertension for more than 10 years, with the highest blood pressure of 180/100 mmHg. Oral valsartan was applied to control blood pressure at 140/80 mmHg. She had no special history of other systematic diseases, tuberculosis, infectious hepatitis, or syphilis. She had no history of surgery, blood transfusion, or drug allergy. She was diagnosed as follows: (I) sepsis, (II) secondary thrombocytopenia, (III) liver abscess, (IV) gram-negative bacilli sepsis (Klebsiella pneumoniae), (V) infection of lumbar vertebrae, (VI) mesenteric panniculitis, (VII) pelvic effusion, (VIII) pericardial effusion, (IX) suppurative endophthalmitis (left), (X) orbital cellulitis (left), (XI) retinal detachment (left), (XII) choroidal detachment (left), (XIII) type 2 diabetes retinopathy (right), (XIV) cortical senile cataract (right, immature stage), (XV) type 2 diabetes, type 2 diabetes nephropathy stage I, and type 2 diabetic peripheral neuropathy, (XVI) hypertension, grade 3 (extremely high risk), (XVII) hepatic cyst, (XVIII) renal cyst, (XIX) hypoproteinemia, (XX) coronary atherosclerotic heart disease, (XXI) lacunar cerebral infarction, (XXII) moderate anemia, and (XXIII) risk of malnutrition.
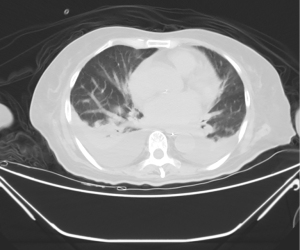
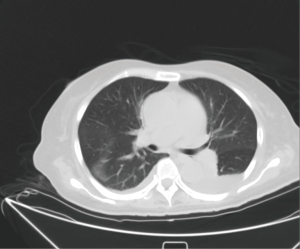
On 20 April, the patient experienced a convulsion with unconsciousness, and she was transferred to the respiratory intensive care unit with pharmaceutical care. An emergency CT examination revealed the lacunar infarction and encephalomalacia; bilateral pleural effusion, and the lower lobe of the right lung was insufficiently inflated, which was slightly more advanced than at the previous examination (Figure 2). She was in a coma and unresponsive, with uncooperative physical examination, slight neck resistance, a weak light response of the eye, uncooperative physical examination of limb muscle strength, low muscle tension, suspicious left Babinski sign (+), and right Babinski sign (−). The impression was “intracranial infection”. Lumbar puncture was performed and the cerebrospinal fluid (CSF) was sent for biochemistry analysis and microbial metagenomic next-generation sequencing (mNGS; IDseqTM Ultra). Biochemistry analysis on 21 April manifested as follows: glucose <1.1 mmol/L ↓, chlorine 108 mmol/L ↓, CSF protein >3,000 mg/L ↑. Microbial mNGS results showed a high sequence of Klebsiella pneumoniae with drug-resistant gene SHV-type beta-lactamases (blaSHV). We administered 2 g meropenem every 8 hours (q8h) prolonged for 3 h to treat intracranial infection, and the body temperature, blood routine, and CRP were gradually improved. The CT examination on 18 May suggested that pulmonary edema and pleural effusion were gradually dissipated and absorbed (Figure 3). The CSF analyses performed on 23 May showed the following: chlorine 119 mmol/L ↓, micro amount of proteins 1,107 mg/L ↑, microalbumin 816.3 mg/L ↑, immunoglobulin G 308.5 mg/L ↑, α2-macroglobulin 18.5 mg/L ↑, and β2-microglobulin 2.67 mg/L ↑. Based on the improvement of clinical manifestations, the patient was discharged from the hospital on 31 May and a close follow-up was conducted. The treatment timeline was shown in Figure 4.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Klebsiella belongs to the Enterobacteriaceae, among which Klebsiella pneumoniae, Klebsiella oxytoca, and Klebsiella ozaenae are inclined to lead to central system infection, and Klebsiella pneumoniae is the most clinically common (10). Here, we have reported an intriguing case of a severe intracranial infection caused by ESBL-producing hvKP in a 65-year-old female. The biochemistry analysis and microbial mNGS of CSF indicated the high sequence of Klebsiella pneumoniae with drug-resistant gene blaSHV. Subsequently, 2 g meropenem q8h prolonged for 3 hours was applied to treat the intracranial infection. The incidence of Klebsiella pneumoniae infection usually develops rapidly with high mortality, herein, we provided this case report of Klebsiella pneumoniae-caused intracranial infection in an adult, and relevant literature was reviewed.
In this case, the patient’s CSF was examined by mNGS and the results suggested the Klebsiella pneumoniae infection with drug-resistant gene blaSHV. The blaSHV gene is named based on its features of hydrolyzing cefothiophene molecule, which belongs to ESBL. Ojdana et al. (11) introduced ESBLs as one of the most well-known resistance mechanisms in gram-negative bacilli. The ESBLs are a group of enzymes that lead to resistance increase in aztreonam, ceftazidime, cefotaxime, related oxyimino-β-lactams, cephalosporins, and penicillins (12). In this case, blaSHV was reported as 1 of the 3 main types of ESBLs. Pishtiwan et al. (13) found that blaSHV accounts for 16.2% in ESBL-producing E. coli strains. Bourgeoning studies have confirmed the presence of blaSHV in Klebsiella pneumoniae infection. Carvalho et al. (14) reported that different ESBL variants of CTX-M and SHV-type were detected in Klebsiella pneumoniae isolates, in occasions associated with carbapenemase genes. Chaudhry et al. (15) found that the highest percentage prevalence of ESBL-producing Klebsiella pneumoniae was wastewater in the hospital, and blaSHV was found at a percentage prevalence of 6%. Therefore, strict rules and regulations should be adopted at the public- as well as hospital-level to restrict the dissemination of antibiotic resistance from the hospital environment to humans.
Carbapenems are the last defensive line to treat gram-negative bacteria with ESBL production (16). However, evidence of hvKP has been increasingly reported in recent years in China, which poses a great threat to clinical treatment (17,18). In this case, for the patient, who had a recurrent fever for more than 1 month, the CT results and laboratory results indicated the systematic infection, and blood culture confirmed the infection of Klebsiella pneumoniae. Although a combination of antibiotics was given, her body temperature remained high, with obvious infectious manifestations and signs detected by laboratory tests and CT examination. Further, she presented with the typical symptoms of intracranial infection, following a physical examination, CT and CSF analyses confirmed the diagnosis of intracranial infection by Klebsiella pneumoniae. At first, we speculated that this Klebsiella pneumoniae strain may belong to hvKP, which acquires drug resistant genes during the disease development. To confirm our hypothesis, CSF microbial mNGS was employed. Unexpectedly, the mNGS results suggested the high sequence of Klebsiella pneumoniae with drug-resistant gene blaSHV, thus we deduced that there may be the coexistence of drug resistance genes and high virulence in Klebsiella pneumoniae in the present case.
It is well accepted that Klebsiella pneumoniae has the potential to develop an increased antimicrobial resistance and virulence. The classic non-virulent strain of ESBL-producing Klebsiella pneumoniae is related to some nosocomial infections (19). Some hvKP strains are associated with invasive infection among previously healthy ambulatory patients, and most of them exhibit antimicrobial susceptibility (20). Several cases of infectious diseases caused by Klebsiella pneumoniae have been reported, which could produce ESBL worldwide. Khaertynov et al. (21) reported the case of a 12-day-old neonate with pyogenic meningitis with poor prognosis, the blood and CSF cultures were positive for Klebsiella pneumoniae, producing ESBL. Klebsiella pneumoniae isolates were resistant to aminopenicillins and third generation cephalosporins, but were sensitive to imipenem and meropenem.
Here, we report a case of adult intracranial infection caused by ESBL-producing hvKP in a 65-year-old female. Biochemistry analysis and microbial mNGS of CSF indicated the high sequence of Klebsiella pneumoniae with drug-resistant gene blaSHV. She was treated with 2 g meropenem q8h prolonged for 3 hours, and the manifestations were relieved. This case highlighted that the outcome of the disease caused by Klebsiella pneumoniae infection is firmly related to the phenotypic features, for instance, virulence factors and antibiotic susceptibility. Here, the presence of ESBL-producing hvKP could contribute to a more severe threat to public health, the appropriate management of similar case such as urgent and rational prevention and treatment is needed.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://atm.amegroups.com/article/view/10.21037/atm-22-3805/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://atm.amegroups.com/article/view/10.21037/atm-22-3805/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wyres KL, Lam MMC, Holt KE. Population genomics of Klebsiella pneumoniae. Nat Rev Microbiol 2020;18:344-59. [Crossref] [PubMed]
- Paczosa MK, Mecsas J. Klebsiella pneumoniae: Going on the Offense with a Strong Defense. Microbiol Mol Biol Rev 2016;80:629-61. [Crossref] [PubMed]
- Li Y, Hu D, Ma X, et al. Convergence of carbapenem resistance and hypervirulence leads to high mortality in patients with postoperative Klebsiella pneumoniae meningitis. J Glob Antimicrob Resist 2021;27:95-100. [Crossref] [PubMed]
- Russo TA, Marr CM. Hypervirulent Klebsiella pneumoniae. Clin Microbiol Rev 2019;32:e00001-19. [Crossref] [PubMed]
- Peng C, Feng DH, Zhan Y, et al. Molecular Epidemiology, Microbial Virulence, and Resistance of Carbapenem-Resistant Enterobacterales Isolates in a Teaching Hospital in Guangzhou, China. Microb Drug Resist 2022;28:698-709. [Crossref] [PubMed]
- Nakamura I, Yamaguchi T, Miura Y, et al. Transmission of extended-spectrum β-lactamase-producing Klebsiella pneumoniae associated with sinks in a surgical hospital ward, confirmed by single-nucleotide polymorphism analysis. J Hosp Infect 2021;118:1-6. [Crossref] [PubMed]
- Wilkowski P, Gajko K, Marczak M, et al. Clinical Significance of Gastrointestinal Carriage of Klebsiella Pneumoniae-Producing Extended-Spectrum Beta-Lactamases in Kidney Graft Recipients. Transplant Proc 2018;50:1874-7. [Crossref] [PubMed]
- Hu Y, Liu C, Shen Z, et al. Prevalence, risk factors and molecular epidemiology of carbapenem-resistant Klebsiella pneumoniae in patients from Zhejiang, China, 2008-2018. Emerg Microbes Infect 2020;9:1771-9. [Crossref] [PubMed]
- Arato V, Raso MM, Gasperini G, et al. Prophylaxis and Treatment against Klebsiella pneumoniae: Current Insights on This Emerging Anti-Microbial Resistant Global Threat. Int J Mol Sci 2021;22:4042. [Crossref] [PubMed]
- Borbone S, Cascone C, Santagati M, et al. Bactericidal activity of ertapenem against major intra-abdominal pathogens. Int J Antimicrob Agents 2006;28:396-401. [Crossref] [PubMed]
- Ojdana D, Sacha P, Wieczorek P, et al. The occurrence of blaCTX-M, blaSHV, and blaTEM genes in extended-spectrum β-lactamase-positive strains of Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis in Poland. International Journal of Antibiotics 2014;2014:935842. [Crossref]
- Livermore DM, Canton R, Gniadkowski M, et al. CTX-M: changing the face of ESBLs in Europe. J Antimicrob Chemother 2007;59:165-74. [Crossref] [PubMed]
- Pishtiwan AH, Khadija KM. Prevalence of blaTEM, blaSHV, and blaCTX-M Genes among ESBL-Producing Klebsiella pneumoniae and Escherichia coli Isolated from Thalassemia Patients in Erbil, Iraq. Mediterr J Hematol Infect Dis 2019;11:e2019041. [Crossref] [PubMed]
- Carvalho I, Carvalho JA, Martínez-Álvarez S, et al. Characterization of ESBL-Producing Escherichia coli and Klebsiella pneumoniae Isolated from Clinical Samples in a Northern Portuguese Hospital: Predominance of CTX-M-15 and High Genetic Diversity. Microorganisms 2021;9:1914. [Crossref] [PubMed]
- Chaudhry TH, Aslam B, Arshad MI, et al. Occurrence of ESBL-producing Klebsiella pneumoniae in hospital settings and waste. Pak J Pharm Sci 2019;32:773-8. [PubMed]
- Jernigan JA, Hatfield KM, Wolford H, et al. Multidrug-Resistant Bacterial Infections in U.S. Hospitalized Patients, 2012-2017. N Engl J Med 2020;382:1309-19. [Crossref] [PubMed]
- Zhang Y, Jin L, Ouyang P, et al. Evolution of hypervirulence in carbapenem-resistant Klebsiella pneumoniae in China: a multicentre, molecular epidemiological analysis. J Antimicrob Chemother 2020;75:327-36. [Crossref] [PubMed]
- Liu C, Shi J, Guo J. High prevalence of hypervirulent Klebsiella pneumoniae infection in the genetic background of elderly patients in two teaching hospitals in China. Infect Drug Resist 2018;11:1031-41. [Crossref] [PubMed]
- Khaertynov KS, Anokhin VA, Rizvanov AA, et al. Virulence Factors and Antibiotic Resistance of Klebsiella pneumoniae Strains Isolated From Neonates With Sepsis. Front Med (Lausanne) 2018;5:225. [Crossref] [PubMed]
- Choby JE, Howard-Anderson J, Weiss DS. Hypervirulent Klebsiella pneumoniae - clinical and molecular perspectives. J Intern Med 2020;287:283-300. [Crossref] [PubMed]
- Khaertynov KS, Anokhin VA, Davidyuk YN, et al. Case of Meningitis in a Neonate Caused by an Extended-Spectrum-Beta-Lactamase-Producing Strain of Hypervirulent Klebsiella pneumoniae. Front Microbiol 2017;8:1576. [Crossref] [PubMed]
(English Language Editor: J. Jones)


