EML4-ALK rearrangement of lung large cell neuroendocrine carcinoma: a case report
Highlight box
Key findings
• The first-line use of targeted therapy is also a good choice for locally advanced L-LCNEC patients with gene mutations.
What is known and what is new?
• For locally advanced L-LCNEC patients with gene mutations, targeted therapy is another choice except chemotherapy for the first-line.
• The first-line use of targeted therapy for L-LCNEC patients with gene mutations is safe and effective, and the quality of life is improved.
What is the implication, and what should change now?
• For patients who are suitable for targeted therapy, first-line targeted therapy is also a good choice not just chemotherapy.
Introduction
Lung large cell neuroendocrine carcinoma (L-LCNEC) is a type of neuroendocrine tumor (NET). It is a rare, poorly differentiated, and highly malignant lung tumor. It accounts for <3% of all lung malignancies (1). L-LCNEC is defined as a non-small cell lung cancer (NSCLC). It is characterized by invasion, extensive metastasis, and a low survival rate, and its clinical characteristics and prognosis are similar to those of small cell lung cancer (SCLC) (2). Because it is very rare, at present, there are still many controversies about its diagnosis, treatment and prognosis, and the relevant treatment and prognosis indicators lack large sample research and literature data support.
A large number of gene mutations have been shown to drive the malignant transformation and cell proliferation of a range of human cancer types, including pulmonary cancer. Anaplastic lymphoma kinase (ALK) gene rearrangement occurs in ~5% of pulmonary carcinomas and is a biomarker for targeted therapy (3-5). To date, several ALK inhibitors, including alectinib, have been developed and used as alternative or first-line options for patients with disease resistant to platinum doublet chemotherapy (6,7). Genomic abnormalities are most frequently observed in L-LCNEC, followed by carcinoid tumor and SCLC (8). Nevertheless, because of L-LCNEC is low prevalence, the molecular characteristics and treatment implications are still unclear. To date, only a few cases of L-LCNEC patients with ALK rearrangements have been reported (9-14); however, the treatment methods used for these patients have differed, and few patients have received a first-line treatment with tyrosine kinase inhibitors. In this article, we report the case of a patient with locally advanced L-LCNEC with an ALK mutation, who received first-line alcetinib targeted therapy that showed good efficacy. We present the following article in accordance with the CARE reporting checklist (available at https://atm.amegroups.com/article/view/10.21037/atm-22-6062/rc).
Case presentation
A previously healthy, 46-year-old, non-smoking woman attended Shenzhen Nanshan District Shekou People’s Hospital for treatment due to cough and expectoration after a cold on November 18, 2020. Chest computed tomography (CT) revealed a nodule in the right lobe (no specific report was available). The doctor at the hospital suggested hospitalization for further diagnosis and treatment, but the patient was scared and refused. She then sought treatment with traditional Chinese medicine. On June 13, 2021, she visited the same hospital for the same reason, and another chest CT revealed that compared to the CT on November 18, 2020, there were more lesions in the upper lobe of the right lung than before, the mediastinal lymph nodes were significantly enlarged, and peripheral lung cancer was likely.
The patient was hospitalized on June 16, 2021. On June 17, 2021, chest CT revealed a 19×16×30 mm nodular shadow in the anterior segment of the upper lobe of the right lung, adjacent pleural involvement, and multiple enlarged lymph nodes were observed at 2R and 4R/L (Right/Left ) in the right hilar and mediastinum. The short diameter of the larger lymph nodes was about 21 mm. The lumen of the superior vena cava was narrowed by lymph nodes pushing, and lymph node metastasis was considered (Figure 1). On June 21, 2021, an ultrasonic bronchoscopy was performed and multiple hypoechoic masses were found in the 4R, 4L, and 7 areas of the mediastinum, right hilar (10R) and right interlobular (11R). The maximum short diameter of the lesion was about 20.4×30.6 mm, in area 4R (Figure 2).


Endobronchial ultrasound–guided transbronchial needle aspiration was performed with a 21 g biopsy needle in the 11R and 4R regions of the mediastinum. On June 22, 2021, the pathological results showed poorly differentiated cancers in the 11R lymph node and 4R lymph node, combined with immunophenotype, conforming to neuroendocrine carcinoma and tending to L-LCNEC.
The immunohistochemistry (IHC) results were positive for thyroid transcription factor 1 (TTF-1), cytokeratin-7 (CK7), AE1/AE3, neural cell adhesion molecule (NCAM, CD56), Chromogranin-A (ChrA), Synaptophysin (Syn), and 40% for Ki-67, but negative for NapsinA, P40, and P63 (Figure 3). The clinical stage was cT1cN3M0 IIIB. Next generation sequencing (NGS) with a 520-gene panel of the patient’ plasma and tumor specimen showed echinoderm microtubule-associated protein-like 4 (EML-4) (exon 13)-ALK (exon 20) fusion with a mutation frequency of 14.48% and 15.37%, microsatellite stability (MSS), tumor proportion score (TPS) 0%, combined positive score (CPS) 1.

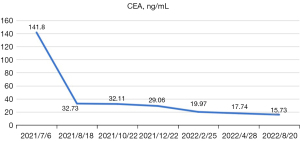
After the evaluation of thoracic tumor surgeon, the doctor suggested that preoperative neoadjuvant chemotherapy should be provided for 2–4 cycles, and a comprehensive assessment should be made to determine whether surgical resection would be possible. The patient refused chemotherapy for personal reasons, and on July 18, 2021, according to the 2020 guidelines of the Chinese Society of Clinical Oncology (CSCO), the patient began treatment with alectinib, an ALK tyrosine kinase inhibitor, at a dosage of 600 mg twice daily. After 1 month of treatment with alectinib, the lung tumor shrank in size from 19×16×30 to 13×11×14 mm, the largest hilar and mediastinal lymph nodes shrank in size from 21 to 14 mm, and the pressure on the superior vena cava also decreased (Figure 1). According to the Response Evaluation Criteria in Solid Tumors (RECIST1.1) evaluation standards, the tumor response was considered a partial response (PR). The patient was then examined every 2 months, and the tumor continued to shrink. On June 24, 2022, Positron emission tomography (PET)-CT was performed and there are still residual tumors, the size of tumors was same to April 28, 2022. On Aug 29, 2022, 12 months after treatment with alectinib, the lung tumor had shrunk in size (compared to chest CT on April 28, 2022) to 9×6 mm, and the short diameter of the hilar and mediastinal lymph nodes was 8 mm, and the level of carcinoembryonic antigen (CEA) continued to decline (Figure 4).
The treatment-related adverse events included skin rashes, which were observed after half a month of medication. The skin rashes emerged on the patient’s hands, feet, and limbs. According to NCI-CTCAE 5.0, the rashes were grades 2–3. The patient received symptomatic treatment with antiallergy Loratadine tablets, skin care, oral and external Chinese medicine, and her skin peeled off after 1 month later. At the time of reexamination in August 2021, the patient’s rash had largely disappeared, and while some of her skin remained pigmented (Figure 5), the skin color had mostly recovered to normal after 6 months of medication. The overall tolerance was acceptable, no other adverse reactions were observed, and the patient was able to live and work normally. The timeline of the case was demonstrated in Figure 6.


All procedures performed in this study were performed in accordance with the ethical standards of the institutional research committee and with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for the publication of this case report and the accompanying images. A copy of the written consent form is available for review by the editorial office of this journal.
Discussion
L-LCNEC, which is a type of NSCLC, is a rare but highly invasive lung cancer. Travis et al. (15) first identified it as a new subtype of lung cancer. In 2011, the World Health Organization classified L-LCNEC as a large cell carcinoma (16). In 2015, it was reclassified as a pulmonary NET together with SCLC, typical carcinoid tumor and atypical carcinoid tumor (17). About 20% of L-LCNEC has a mixed tumor component with SCLC or NSCLC (17). To study the biological relationship between L-LCNEC and SCLC and NSCLC, Rekhtman et al. (18) analyzed the genomic changes of L-LCNEC and other major lung cancer types and found that L-LCNEC mainly comprised the following 2 distinct subgroups characterized by different genomic signatures: the SCLC-like subtype (characterized by a TP53+RB1 co-mutation/loss and other SCLC-type alterations); and the NSCLC-like subtype (characterized by a lack of co-altered TP53+RB1 and the nearly universal occurrence of the NSCLC-type mutations of STK11, KRAS, and KEAP1).
Research has been conducted to determine what the biological behavior and prognosis of L-LCNEC is most like. Some people have suggested that L-LCNEC has neuroendocrine characteristics and should be considered a small cell carcinoma (19). To date, there have been no randomized controlled trials examining the optimal treatment for L-LCNEC. Derks et al. (20) conducted a retrospective study and found that L-LCNEC is a highly aggressive lung cancer like SCLC with a poor prognosis at all stages of the disease. However, there are important differences between L-LCNEC and SCLC. Notably, in stage IV, the prognosis of L-LCNEC patients is worse than that of SCLC patients; however, the treatment currently used to treat early stage L-LCNEC differs to that used to treat SCLC. Some studies have shown that in advanced stage L-LCNEC, the objective response rate of platinum-based chemotherapy regimens for SCLC (37–52%) is higher than that of NSCLC regimens (12–50%). However, patients’ responses are usually transient, with a median progression-free survival (PFS) time of 4.6–6.1 months, and a poor median overall survival (OS) time of 10.2–11.1 months (19,21). Another study showed that patients with stage IIIB and IV L-CNEC who undergo chemotherapy have unfavorable outcomes, with a median PFS time of 5.2 months and OS time of 5.2 months (22).
The main characteristic of L-LCNEC IHC is the expression of neuroendocrine related markers, such as CD56, CgA, and Syn, which can be used as markers for the pathological differential diagnosis of NSCLC (23). In this article, we reported a rare case in which the patient was diagnosed with neuroendocrine carcinoma, which was likely to be large cell neuroendocrine carcinoma. The IHC results for a number of neuroendocrine markers, such as CD56, ChrA, and Syno, were positive.
Gene mutations in tyrosine kinases play essential roles in the pathogenesis of lung adenocarcinoma. Tumors harboring an ALK rearrangement can be successfully treated with ALK inhibitors. ALK rearrangement is most commonly observed in lung adenocarcinoma (24).
L-LCNEC harboring an ALK rearrangement is very rare, and at present, no large-scale clinical trials have been conducted on this disease. However, it has been reported that ALK-positive tumors usually express TTF-1 (25). In the present case, the patient was TTF-1 positive (3+); however, after communication with the pathology department of our hospital, it was confirmed that the patient had simple L-LCNEC. Moreover, the TP53 + RB1 co-mutation was not detected in the patient’s genetic test results, and it is speculated that the patient had a NSCLC-like subtype.
At present, the tumor treatment mode is more individualized and precise than decades. The guidelines on NSCLC issued by the CSCO and the National Comprehensive Cancer Network both recommend that patients undergo polygenic testing to clarify their molecular typing. NGS was conducted to detect the rearrangement (fusion) of EML4-ALK (E13:A20) in our patient, which is the most common rearrangement (fusion) site. In recent years, several cases have reported that L-LCNEC has the ALK rearrangement. Indeed, the 2 earliest reported cases of L-LCNEC reported ALK rearrangement. The first case involved a 43-year-old female who had never smoked and received targeted treatment with crizotinib at the first line. After 6 weeks of treatment, she developed brain metastasis and skin metastasis, indicating that the tumor was resistant to crizotinib (9). The second case was a 60-year-old female who had never smoked and did not receive any ALK inhibitors (10). Additionally, Hayashi et al. (11) reported the case of a 75-year-old, non-smoking, female diagnosed with L-LCNEC with multiple liver and bone metastases. After 7 cycles of cytotoxic drug chemotherapy, her genotype test showed ALK rearrangement. She subsequently took alectinib. About 1 month later, the tumor markers neuron-specific-enolase (NSE) and pro-gastrin-releasing peptide (ProGRP) decreased to the normal range, and 4 months later, the lung lesions were reduced from 21 to 16 mm, the liver lesions regressed, and the patient achieved PR. After 6 months of treatment, positron emission tomography–CT showed that the disease was stable.
The patient in the present case was in the cT1cN3M0 IIIB clinical stage, and there was no surgical indication at that time. The patient refused chemotherapy for personal reasons, and demanded targeted therapy.
Based on the results of Phase III clinical study of ALESIA (26). In 2018, China’s National Medical Products Administration approved alectinib for the treatment of ALK-positive locally advanced or metastatic NSCLC, including its use as a first-line and second-line drug after the treatment of progression of crizotinib. As the median PFS time of the first-line treatment of alectinib is 34.8 months, the CSCO guidelines recommend it as the first-line treatment for ALK-positive patients.
In the present case, after 1 month of targeted treatment with alectinib, the tumor was significantly reduced, and the patient achieved PR. The tumor was then regularly examined every 2 months, and continued to shrink. After 1 year of treatment, the tumor was reviewed, and the PR status was maintained. In the initial stage of treatment, a grade 2–3 rash appeared, but after treatment, the rash quickly improved and did not re-appear. No other adverse reactions were observed and the patient was able to live and work normally.
A previous study has shown that palliative chemotherapy for SCLC is more suitable than NSCLC for advanced L-LCNEC (20). Immunotherapy has also been tried in the treatment of L-LCNEC (1,27-29). For patients who are suitable for targeted therapy, first-line targeted therapy is also a good choice, and more trials and studies to explore the best and appropriate treatment of L-LCNEC are needed in the future. In this case, it is a pity that ALK immunohistochemical test was not carried out for the patient initially, and we will improve the relevant immunohistochemistry later.
Conclusions
For NSCLC patients with unresectable stage III and a performance status of 0-1, it is recommended to conduct multidisciplinary team discussion, radical concurrent radiochemotherapy or Duvalizumab should be used as the consolidation treatment after concurrent radiochemotherapy as the Level I recommendation of the 2020 COSO Guidelines. Targeted treatment is preferred for patients with stage IV drive gene positive NSCLC. But targeted treatment is also a good choice for patients with stage III who refuse radiochemotherapy and have gene mutations, in this case, the patient chose targeted therapy and refused chemotherapy and surgery, she has obtained good curative effect without obvious serious side effects and improved the quality of life.
Acknowledgments
We would like to thank the patient, her family, and all the investigators involved in this study.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://atm.amegroups.com/article/view/10.21037/atm-22-6062/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://atm.amegroups.com/article/view/10.21037/atm-22-6062/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work, including ensuring that any questions related to the accuracy or integrity of any part of the work have been appropriately investigated and resolved. All procedures performed in this study were performed in accordance with the ethical standards of the institutional research committee and with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for the publication of this case report and the accompanying images. A copy of the written consent form is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Mauclet C, Duplaquet F, Pirard L, et al. Complete tumor response of a locally advanced lung large-cell neuroendocrine carcinoma after palliative thoracic radiotherapy and immunotherapy with nivolumab. Lung Cancer 2019;128:53-6. [Crossref] [PubMed]
- Asamura H, Kameya T, Matsuno Y, et al. Neuroendocrine neoplasms of the lung: a prognostic spectrum. J Clin Oncol 2006;24:70-6. [Crossref] [PubMed]
- Soda M, Choi YL, Enomoto M, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 2007;448:561-6. [Crossref] [PubMed]
- Paik JH, Choe G, Kim H, et al. Screening of anaplastic lymphoma kinase rearrangement by immunohistochemistry in non-small cell lung cancer: correlation with fluorescence in situ hybridization. J Thorac Oncol 2011;6:466-72. [Crossref] [PubMed]
- Perner S, Wagner PL, Demichelis F, et al. EML4-ALK fusion lung cancer: a rare acquired event. Neoplasia 2008;10:298-302. [Crossref] [PubMed]
- Seto T, Kiura K, Nishio M, et al. CH5424802 (RO5424802) for patients with ALK-rearranged advanced non-small-cell lung cancer (AF-001JP study): a single-arm, open-label, phase 1-2 study. Lancet Oncol 2013;14:590-8. [Crossref] [PubMed]
- Shaw AT, Kim DW, Mehra R, et al. Ceritinib in ALK-rearranged non-small-cell lung cancer. N Engl J Med 2014;370:1189-97. [Crossref] [PubMed]
- Lou G, Yu X, Song Z. Molecular Profiling and Survival of Completely Resected Primary Pulmonary Neuroendocrine Carcinoma. Clin Lung Cancer 2017;18:e197-201. [Crossref] [PubMed]
- Omachi N, Shimizu S, Kawaguchi T, et al. A case of large-cell neuroendocrine carcinoma harboring an EML4-ALK rearrangement with resistance to the ALK inhibitor crizotinib. J Thorac Oncol 2014;9:e40-2. [Crossref] [PubMed]
- Kato M, Higuchi K, Shimojo HA, et al. Cytology of large- cell neuroendocrine carcinoma harboring an EML 4-ALK rearrangement-a case report. Medical Journal of Aizawa Hospital 2015;13:57-63.
- Hayashi N, Fujita A, Saikai T, et al. Large Cell Neuroendocrine Carcinoma Harboring an Anaplastic Lymphoma Kinase (ALK) Rearrangement with Response to Alectinib. Intern Med 2018;57:713-6. [Crossref] [PubMed]
- Fu Z, Zhu G, Wang L, et al. Case Report: A Pregnant Woman Diagnosed as ALK-Rearrangement Lung Large Cell Neuroendocrine Cancer With Brain Metastasis. Front Oncol 2022;12:823813. [Crossref] [PubMed]
- Akhoundova D, Haberecker M, Fritsch R, et al. Targeting ALK in Neuroendocrine Tumors of the Lung. Front Oncol 2022;12:911294. [Crossref] [PubMed]
- Lim CA, Banyi N, Tucker T, et al. A Case of ALK-Rearranged Combined Lung Adenocarcinoma and Neuroendocrine Carcinoma with Diffuse Bone Metastasis and Partial Response to Alectinib. Curr Oncol 2022;29:848-52. [Crossref] [PubMed]
- Travis WD, Linnoila RI, Tsokos MG, et al. Neuroendocrine tumors of the lung with proposed criteria for large-cell neuroendocrine carcinoma. An ultrastructural, immunohistochemical, and flow cytometric study of 35 cases. Am J Surg Pathol 1991;15:529-53. [Crossref] [PubMed]
- Varlotto JM, Medford-Davis LN, Recht A, et al. Should large cell neuroendocrine lung carcinoma be classified and treated as a small cell lung cancer or with other large cell carcinomas? J Thorac Oncol 2011;6:1050-8. [Crossref] [PubMed]
- Travis WD, Brambilla E, Nicholson AG, et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol 2015;10:1243-60. [Crossref] [PubMed]
- Rekhtman N, Pietanza MC, Hellmann MD, et al. Next-Generation Sequencing of Pulmonary Large Cell Neuroendocrine Carcinoma Reveals Small Cell Carcinoma-like and Non-Small Cell Carcinoma-like Subsets. Clin Cancer Res 2016;22:3618-29. [Crossref] [PubMed]
- Sun JM, Ahn MJ, Ahn JS, et al. Chemotherapy for pulmonary large cell neuroendocrine carcinoma: similar to that for small cell lung cancer or non-small cell lung cancer? Lung Cancer 2012;77:365-70. [Crossref] [PubMed]
- Derks JL, Hendriks LE, Buikhuisen WA, et al. Clinical features of large cell neuroendocrine carcinoma: a population-based overview. Eur Respir J 2016;47:615-24. [Crossref] [PubMed]
- Naidoo J, Santos-Zabala ML, Iyriboz T, et al. Large Cell Neuroendocrine Carcinoma of the Lung: Clinico-Pathologic Features, Treatment, and Outcomes. Clin Lung Cancer 2016;17:e121-9. [Crossref] [PubMed]
- Le Treut J, Sault MC, Lena H, et al. Multicentre phase II study of cisplatin-etoposide chemotherapy for advanced large-cell neuroendocrine lung carcinoma: the GFPC 0302 study. Ann Oncol 2013;24:1548-52. [Crossref] [PubMed]
- Metovic J, Barella M, Bianchi F, et al. Morphologic and molecular classification of lung neuroendocrine neoplasms. Virchows Arch 2021;478:5-19. [Crossref] [PubMed]
- Shaw AT, Kim DW, Nakagawa K, et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N Engl J Med 2013;368:2385-94. [Crossref] [PubMed]
- Yoshida A, Tsuta K, Watanabe S, et al. Frequent ALK rearrangement and TTF-1/p63 co-expression in lung adenocarcinoma with signet-ring cell component. Lung Cancer 2011;72:309-15. [Crossref] [PubMed]
- Zhou C, Lu Y, Kim SW, et al. Primary results of ALESIA: phase III, randomised open-label study of alectinib(ALC) vs crizotinib(CRZ) in Asian patients(pts) with treatment-naive ALK plu advanced non-small-cell lung cancer(NSCLC). Ann Oncol 2018;29:174.
- Sherman S, Rotem O, Shochat T, et al. Efficacy of immune check-point inhibitors (ICPi) in large cell neuroendocrine tumors of lung (LCNEC). Lung Cancer 2020;143:40-6. [Crossref] [PubMed]
- Chaft JE, Hellmann MD, Velez MJ, et al. Initial Experience With Lung Cancer Resection After Treatment With T-Cell Checkpoint Inhibitors. Ann Thorac Surg 2017;104:e217-8. [Crossref] [PubMed]
- Chauhan A, Arnold SM, Kolesar J, et al. Immune checkpoint inhibitors in large cell neuroendocrine carcinoma: current status. Oncotarget 2018;9:14738-40. [Crossref] [PubMed]