
An interventional pilot study protocol on the effect of extra virgin olive oil on women with preeclampsia risk
Introduction
Background
Preeclampsia (PE), a multisystem and complex disorder diagnosed when maternal hypertension manifests after 20 weeks of gestation with proteinuria, is one of the direct causes of maternal morbidity and mortality in addition to bleeding and infection (1). The global incidence is around 2–8% of total pregnancies (2) and may reach 30% in the high-risk population (3). The clinical manifestations of PE are hypertension and proteinuria, which indicate disorders of the microvascular system, namely vasoconstriction and increased vascular permeability (4). Severe cases are also associated with high rates of perinatal death, prematurity, and newborns that are small for gestational age (SGAs) (5).
Previous studies showed that oxidative stress, defined as an imbalance between oxidants and antioxidants, had an important role in the pathophysiology of PE (6). Reduced perfusion and ischemic reperfusion in the placenta result in placental hypoxia and raised synthesis of free radicals in the placenta, leading to vasoconstriction and microvascular damage (7). Some authors suggested antioxidant supplementation, such as vitamin E and C, in order to decrease vascular and endothelial damage (8). Others showed a non-significant effect of vitamin D status (VDS) in preventing PE (9); however, issues of dose, timing, and duration of supplementation have not been completely addressed (10). Moreover, it was postulated that VDS augmentation at earlier time-points of pregnancy, rather than the third trimester, may be necessary to maintain sufficient vitamin D levels protective against PE (10).
Although the etiology of PE has not been elucidated, several clinical risk factors for this pathology have been reported, including primiparity, advanced maternal age, previous PE, multiple gestations, high body mass index (BMI) and preexisting diseases like chronic hypertension, antiphospholipid antibody syndrome and chronic renal diseases (11,12). In a recent study, a 2 h oral glucose tolerance test (OGTT) had a significant impact on blood pressure and lipid profile determinants in Middle Eastern pregnant women with gestational diabetes mellitus (GDM) and PE (13). It was also suggested that glucose intolerance during pregnancy, reflected by elevated results of a 50 g 1 h post-oral glucose challenge test (GCT), is associated with gestational hypertension and hyperlipidemia as a risk factor for PE (14). Treatments of GDM before 20 weeks’ gestation led to modestly fewer adverse neonatal outcomes (15), and professional societies recommend low-dose aspirin for preventing PE in high-risk women (16); however, focused and efficient dietary treatments as a protentional treatment are scarce (16,17).
Rationale and knowledge gap
Extra virgin olive oil (EVOO) is considered a rich source of antioxidant compounds and is a central food in the Mediterranean diet (18). Moreover, it was recently indicated that EVOO from early harvested olives (EVOOEH) was more concentrated with antioxidant compounds in different olive types and regions (19,20). It is possible that EVOO ingredients such as polyphenols act in human body as an antioxidant by inhibiting nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (21). Such NADPH oxidase constrain, can reduce oxidative stress and protect the vascular endothelium by lowering reactive oxygen species (ROS) production, repairing (21,22). This can also enhances endothelial nitric oxide (NO) bioavailability, promoting vasodilation and lowering blood pressure (22). EVOO’s polyphenols also attenuate endothelin-1 (ET-1), a vasoconstrictor, further supporting blood pressure reduction (23). This may be important at gestational weeks 8–18, when spiral artery remodeling occur—a critical time window for normal placentation (23). In animal models, EVOO has been shown to modulate angiogenic function by increasing vascular endothelial growth factor (VEGF) and decreasing soluble FMS-like tyrosine kinase 1 (sFlt-1) (21). It was recently reported that an EVOO consumption of 36 g/d (three tablespoons) from gestational weeks 24–28 until term delivery among GDM pregnant women was associated with a lower prevalence of maternal hypertriglyceridemia and has anti-inflammatory properties in placenta and umbilical cord blood. This was suggested in a clinical trial involving Mediterranean diet with additional EVOO and pistachios that resulted in decreased incidence of GDM (24). Another report showed that EVOO consumption >40 mL/d from gestational weeks 12–14 until delivery among healthy pregnant women was associated with a lower prevalence of GDM (25). Thus, our hypotheses are that EVOOEH consumption can decrease microvascular damage and poor placentation at the first half of pregnancy, as well as dysglycemia thorough its enhanced anti-oxidative activity. Also, we postulate that higher VDS (within the adequate dietary range for pregnancy) can decrease the odds for GDM, a PE risk factor (14).
Study protocol objective
To explore the role of EVOOEH in reducing PE risk.
Specific aims:
- To evaluate the effect of EVOOEH interventions on PE incidence during the entire pregnancy.
- To assess the impact of these interventions on GDM and PE-related markers and other adverse pregnancy outcomes in the high-risk population for PE.
We present this article in accordance with the SPIRIT reporting checklist (available at https://atm.amegroups.com/article/view/10.21037/atm-25-122/rc).
Methods
Study design
- Study type: interventional;
- Estimated enrollment: 156 participants;
- Allocation: randomized;
- Intervention model: parallel assignment;
- Intervention model description: dietary intervention study: high-risk pregnant women receiving the Ministry of Health (MOH) general dietary recommendations for pregnancy (MOH recommendations) with addition of three tablespoons (42 mL) EVOOEH per day at each morning for four weeks during the first half of pregnancy (gestational ages between 8 to 16 weeks);
- Compliance markers: a 17-point Mediterranean Diet Adherence Screener adapted to Israel (I-MEDAS) (26) score will be evaluated by a registered dietitian (RD) and maternal whole blood and plasma hydroxytyrosol (HT) (at recruitment and ~ four weeks post-intervention initiation);
- Masking: single (investigator);
- Masking description: data analysis will be masked;
- Primary purpose: prevention.
Detailed enrolment, interventions and assessments schedule in Table 1.
Table 1
| Time point | Enrollment (GA 8–16 weeks) | Intervention w 1 | Intervention w 2 | Intervention w 3 | Intervention w 4 (end, GA 12–20 weeks) | Follow-up (ongoing to delivery, ~GA 40 weeks) |
|---|---|---|---|---|---|---|
| Enrollment activities | Informed consent. Randomization†. Baseline blood sample [sFlt-1/PlGF, 25(OH)D, HT]. Provide Links (MOH Recs, I-MEDAS questionnaire). Start aspirin (if indicated during GA 12-28w) | |||||
| Interventions | EVOOEH arm: 42 mL/day EVOOEH + MOH Recs | Continue daily intervention | Continue daily intervention | End 4w intervention | Standard care + aspirin if indicated | |
| Control arm: MOH Recs only | Monitor outcomes throughout pregnancy | |||||
| Assessments/visits | In-person visit: blood draw, I-MEDAS questionnaire | In-person visit: post-intervention blood sample (HT, sFlt-1/PlGF, 25(OH)D). Adherence check by RD | Routine prenatal visits: BP monitoring, GDM diagnosis (if not done), PE assessment | |||
| Delivery: gestational age, newborn weight percentile, CS, preterm, SGA incidence | ||||||
| Outcome measures | Baseline data collection (including demographics and risk factors) | Primary: PE, GDM, preterm and SGA incidence | Secondary: post intervention sFlt-1/PlGF ratio, GCT, BP (SBP/DBP), gestational age at delivery, newborn outcomes | |||
| Adherence | I-MEDAS score | Phone call (~15 min): adherence check by RD | Phone call (~15 min): adherence check by RD | Adherence check by RD and I-MEDAS score |
†, random allocation sequence was generated via NIH Clinical Trial Randomization Tool (link: https://ctrandomization.cancer.gov/tool/). 25(OH)D, serum 25-hydroxyvitamin D; BP, blood pressure; CS, cesarean section; DBP, diastolic blood pressure; EVOOHEH, early harvested extra virgin olive oil; GA, gestational age; GCT, glucose challenge test (which is performed at GA of 24–28 w); GDM, gestational diabetes; HT, hydroxytyrosol; I-MEDAS, Mediterranean Diet Adherence Screener adopted to Israel; MOH Recs, Israeli Ministry of Health general dietary recommendations for pregnancy; NIH, National Institutes of Health (USA); RD, registered dietitian licensed by MOH; SBP, systolic blood pressure; sFlt-1/PlGF, soluble fms-like tyrosine kinase 1 to placental growth factor; SGA, small for gestational age.
Arms and interventions
EVOOEH arm: daily addition of 42 mL (three tablespoons) at each morning of EVOOEH to MOH recommendations for 4 weeks (n=78). Control arm: MOH recommendations for 4 weeks (n=78).
Notes:
- Random arms allocation sequence was generated via the united states National Institutes of Health (NIH) Clinical Trial Randomization Tool (link: https://ctrandomization.cancer.gov/tool/). A single-masking (investigator and data analyst) regarding arms is employed.
- Participant will receive a link to fill out the I-MEDAS questionnaire (link: https://ariel.qualtrics.com/jfe/form/SV_1ZwojTnNaJYe5Ia) and to MOH recommendations (link: https://me.health.gov.il/parenting/family-planning/healthy-pregnancy-and-preventing-risks/nutrition-pregnancy/nutrition-pregnancy/) and a registered dietitian (RD) will make two follow up phone calls (about 15 minutes each) to all participants at the second and fourth week of intervention to ensure adherence for each arm.
- According to MOH recommendations, no restriction regarding EVOO consumption will be highlighted by the RD or other research staff member.
- The intervention will take place along with low-dose aspirin prophylaxis as recommended in women at high risk of PE (normally initiated between 12 weeks and 28 weeks of gestation) by the American College of Obstetrics and Gynecology (ACOG) guidelines, if needed (27).
- Adherence will be objectively quantified via pre- and post-intervention whole blood and plasma HT concentrations, I-MEDAS score (self-report questionnaire), and RD phone calls (weeks 2/4). Deviations will be managed via RD counseling and sensitivity analyses (see Advanced analysis).
- Safety monitoring includes participant self-reported symptoms (graded by CTCAE v5.0), RD recording, and medical file review for AEs per the Barzilai University Medical Center at Ashkelon (BUMCA)’s Helsinki Committee, using these criteria: Gastrointestinal (mild nausea, diarrhea, or dyspepsia onset 1–4 hours post-consumption, resolving within 24–48 hours); Metabolic (gestational weight gain >0.5 kg/week or hypertriglyceridemia >500 mg/dL over 2–4 weeks); Allergic/Dermatologic (rash/pruritus within 30 minutes to two hours after ingestion/application); Serious AEs (hypertension >160/110 mmHg within 1–7 days, triggering unblinding/withdrawal).
- Data management includes coded data in secure de-identified storage (BUMCA servers) and quality assurance per good clinical practice (GCP) guidelines.
Outcome measures
- GDM, PE, Cesarean section (CS), preterm and SGA incidence.
- Maternal systolic and diastolic blood pressure (SBP and DBP, respectively).
- Gestational age at delivery.
- Adjusted newborn weight percentile.
- Post intervention maternal GCT result at the end of intervention.
- Serum sFlt-1/PlGF ratio at the end of intervention.
Independent variables
Independent variables (including known risk factors for PE): demographic (including age), gynecological (including parity and all background diagnoses), obstetrics (including all current pregnancy diagnoses and gravidity), dietary (I-MEDAS score, including self-reported EVOO consumption, as well as levels of 25-hydroxyvitamin D in maternal serum [25(OH)D]), anthropometrics (including height and weight) and others.
Software for data extraction
Software to be used—the BUMCA portfolio and any other data source relevant to participants’ and their newborns’ information needed for this study. In order to protect confidentiality before, during, and after the trial, identifying information will be omitted and each participant will receive a coded serial number before sharing the data.
Eligibility criteria eligibility for enrollment
- Ages: 18 to 45 years.
- Gender: women.
- Exceptions: low risk pregnancy or blindness or illiteracy or experiencing a medical emergency.
Inclusion criteria:
- Pregnant women with clinical risk factors for PE [previous PE, primigravida, chronic hypertension, chronic renal disease, autoimmune disease (e.g., systemic lupus erythematosus, antiphospholipid antibody syndrome, Hashimoto thyroiditis), overweight (BMI >25 kg/m2), obesity (BMI >30 kg/m2), morbid obesity (BMI >40 kg/m2), pregestational diabetes (type 1 or type 2), multiple pregnancy].
- Gestational ages of 8 to 16 weeks BUMCA’s Obstetrics and Gynecologic division.
- Without known or self-reported consumption of EVOO above 28 mL/d (two tablespoons) per day.
Exclusion criteria:
- Infectious diseases.
- Refusing enrollment or participation.
- Do not follow low-dose aspirin prophylaxis (if instructed).
Assays
Blood sampling
To determine 25(OH)D, HT and sFlt-1/PlGF ratio, the participants will provide non-fasting venous blood samples within 24 h of recruitment.
Laboratory analysis and equipment
- 25(OH)D: samples will be centrifuged immediately, and the serum will be separated and stored at −4 ℃ until analysis. The 25(OH)D will be analyzed by the clinical biochemistry laboratory at BUMCA using LIAISON XL analyzer (DiaSorin Inc., Via Crescentino Snc Saluggia 13040, Italy). Reference ranges will be 0.84–6.62 pg/mL and 0.79–5.91 pg/mL for first and second trimesters (respectively) (28).
- HT: samples will be immediately stored at −20 ℃ until centrifugation and plasma separation before analysis. Olive oil phenols and HT, including but not limited to tyrosol, HT, homovanillyl alcohol and their metabolites (sulfate, acetate, etc. as shown in Figure 1), are planned to be analyzed by the Targeted Metabolomics Unit at the Weizmann Institute of Science using a highly sensitive method on state-of-the-art equipment consisting of liquid chromatography coupled with tandem mass spectrometry (UPLC-MS/MS). For this study, the 20 µL of samples 400 µL of ice-cold methanol and 5 µL of HT-d4 (10 µg/mL; internal standard) are added. After intensive vortexing, the mixtures are agitated on a shaker (10 ℃; 1,200 rpm; 40 min; Thermomix C, Eppendorf). Precipitates are separated by centrifugation (6 ℃; 21,000 g; 15 min), and the supernatants are collected, evaporated in Speedvac, and re-dissolved in 100 µL of 10%-aqueous methanol for consequent LC-MS/MS analysis. The LC-MS/MS instrument consisting of an Acuity I-class UPLC system (Waters) and Xevo TQ-S triple quadrupole mass spectrometer (Waters), equipped with an electrospray ion source and operated in negative ion mode, was used for analysis of HT. MassLynx and TargetLynx software (v.4.2, Waters) were applied for the acquisition and analysis of data. Chromatographic separation was performed on a 1.7-µm Acuity UPLC BEH C18 column (100 mm × 2.1 mm internal diameter (Waters), with mobile phases A (0.1% aqueous formic acid, v/v) and B (0.1% of formic acid in acetonitrile, v/v) at a flow rate of 0.3 mL·min−1 and column temperature of 30 ℃. A linear gradient from 0 to 50% B during 2.6 min is used. Samples kept at 10 ℃ are automatically injected in a volume of 2 µL. For MS, argon is used as the collision gas with a flow of 0.10 mL·min−1.
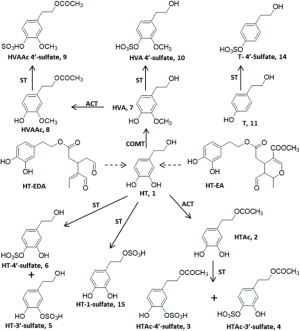 Figure 1 Olive oil polyphenol metabolites that can be detected in human plasma by using UPLC-MS/MS after EVOO. The figure shows the compounds that can be detected and quantified in human plasma (following EVOO consumption) using UPLC-MS/MS by the Targeted Metabolomics Unit at the Weizmann Institute of Science. Arrows may represent a direct or an indirect metabolic pathway. ACT, O-acetyltransferase; COMT, O-catechol methyl transferase; EVOO, extra virgin olive oil; HT, hydroxytyrosol; HTAc, hydroxytyrosol acetate; HVA, homovanillyl alcohol; HVAAc, homovanillyl alcohol acetate; SF, sulfotransferase; T, tyrosol; UPLC-MS/MS, ultra-high performance liquid chromatography coupled with tandem mass spectrometry.
Figure 1 Olive oil polyphenol metabolites that can be detected in human plasma by using UPLC-MS/MS after EVOO. The figure shows the compounds that can be detected and quantified in human plasma (following EVOO consumption) using UPLC-MS/MS by the Targeted Metabolomics Unit at the Weizmann Institute of Science. Arrows may represent a direct or an indirect metabolic pathway. ACT, O-acetyltransferase; COMT, O-catechol methyl transferase; EVOO, extra virgin olive oil; HT, hydroxytyrosol; HTAc, hydroxytyrosol acetate; HVA, homovanillyl alcohol; HVAAc, homovanillyl alcohol acetate; SF, sulfotransferase; T, tyrosol; UPLC-MS/MS, ultra-high performance liquid chromatography coupled with tandem mass spectrometry. - sFlt-1/PlGF ratio: samples are centrifuged immediately at 3,000 ×g for 10 minutes, and the serum is separated and stored at −20 ℃ until analysis, with processing completed within 6 months to ensure stability. The sFlt-1/PlGF ratio will be analyzed by the clinical biochemistry laboratory at BUMCA using Cobas e 601 analyzer (Roche Diagnostic GmbH Sandhofer strasse 116 D-68305 Mannheim) on the Cobas e601 electrochemiluminescence immunoassay analyzer. The assay employs automated sandwich immunoassay principles with specific monoclonal antibodies, yielding results in <18 minutes per sample. Assay validation followed manufacturer protocols and Clinical and Laboratory Standards Institute (CLSI) guidelines, including linearity across 0–10,000 pg/mL for sFlt-1 and 3–2,000 pg/mL for PlGF (r2>0.99), limits of detection/quantification at 6/10 pg/mL (sFlt-1) and 5/7 pg/mL (PlGF), and no significant interference from hemolysis, lipemia, or bilirubin up to clinical thresholds. Reproducibility was verified via intra-assay (CV <3% at low/medium/high controls), inter-assay (CV <5%), and total imprecision (CV <5%) across multiple runs and sites, with quality control using two-level commercial materials run daily.
Preliminary results
Based on our initial literature review and laboratory preparations, the detection of olive oil phenols including HT is feasible, as described in a recently developed method (29), which has also been modified and described in a more recent report (30). Figure 1 shows the compounds that can be detected and quantified in human plasma (following EVOO consumption) using ultra-high-performance liquid chromatography coupled with tandem mass spectrometry (UPLC-MS/MS) by the Targeted Metabolomics Unit at the Weizmann Institute of Science.
Statistical analysis
Sample size
For sample size calculation, the primary endpoint was the incidence of PE during the second half of pregnancy. Assuming a high incidence of 38.5% in the control arm based on a high-risk population (31) vs. typical incidence of 17.1% in the intervention arm based on PE incidence among Israeli women who had PE in their first pregnancy (32). Hence, we estimated that 68 pregnant women would be required per arm to provide statistical power of 80% (2-tailed, α error of 0.05). Given possible 15% loss to follow-up and discontinuation/missing data, we will enroll 78 volunteering pregnant women in each group (N=156), guaranteeing 4-week of intervention completion and the required final sample size for statistical analysis post-exclusions.
Advanced analysis
The statistical analysis will be conducted using JMP Pro software (version 17, SAS Institute, Cary, NC, USA). The dependent variables will be either dichotomous categories (e.g., PE and GDM) or continuous variables [e.g., 25(OH)D and HT concentrations, sFlt-1/PlGF ratio, gestational age at recruitment or birth and birth weight percentile]. Categorical parameters will be summarized by number and percentage and will be compared by arms using the chi-squared test, followed by the Fisher’s exact test and odds ratio (OR) calculation when applicable. For continuous variables, Student’s t-tests or Wilcoxon rank-sum tests will be performed. Pre- and post-intervention whole blood and plasma HT concentrations, the I-MEDAS at baseline and endpoint, and structured RD phone calls at weeks 2 and 4 fulfillment will be compared and tested as continuous variables. Deviations from the protocol will be handled with sensitivity analyses excluding non-adherent participants [e.g., I-MEDAS score or HT concentrations below or above 10% or 90% intra-group distribution (respectively)] to evaluate robustness of primary outcomes. The independent variables will include HT concentrations. Multivariate analyses will include both demographic and clinical characteristics (such as BMI, parity, maternal age) as well as outcomes, if significant, according to the univariate analyses. A two-tailed test with a P value below 0.05 will be considered statistically significant.
Ethics and Dissemination
This study protocol (version number 3.0 from October 10th 2024) has been approved by the Helsinki Committee of Barzilai University Medical Center at Ashkelon (BUMCA) (approval No. 0049-24-BRZ). All participants will provide the recruiting RDs informed written consent before enrollment. The study will be conducted in accordance with the Declaration of Helsinki and its subsequent amendments and Good Clinical Practice guidelines. The findings of this study will be disseminated through peer-reviewed publications and presentations at scientific conferences. We aim to publish the results in a high-impact journal within the fields of obstetrics and gynecology or nutrition.
Achieved study milestones
The study was appropriately reported at the Israeli MOH and appears at its clinical trials online database website named ‘MyTrial’ (33) and the National Institutes of Health (NIH) of the United States government and appears at its clinical trials online database website named ‘ClinicalTrials.gov’ (NCT06759545) (34). Additionally, 14 participants were recruited at the BUMCA’s Obstetrics and Gynecology Division from January 28 to July 28 2025 (without AEs in this period) with estimated calculated recruitment rate of 2–3 participants/month (see flow chart in Figure 2). The baseline characteristics of study participants in the EHEVOO and Control groups are presented in Table 2.

Table 2
| Category | Characteristic | EHEVOO (n=7) | Control (n=7) |
|---|---|---|---|
| Sociodemographic | Tertiary education | 5 (71) | 4 (57) |
| Age (years) | 30 [25.5–36.5] | 30 [26–36] | |
| Anthropometrics | Weight (kg) | 88 [75.7–101.8] | 77 [69.7–97] |
| Height (m) | 1.6 [1.6–1.7] | 1.7 [1.6–1.8] | |
| BMI (kg/m2) | 34 [28–39] | 30 [24–35] | |
| Clinical background | Previous preeclampsia | 0 (0) | 0 (0) |
| Chronic hypertension | 0 (0) | 0 (0) | |
| CKD | 0 (0) | 0 (0) | |
| T2DM | 0 (0) | 1 (17) | |
| T1DM | 0 (0) | 0 (0) | |
| AID | 2 (28) | 2 (28) | |
| IVF pregnancy | 2 (28) | 2 (28) | |
| Initial self-reported habits | Current smoker | 1 (14) | 2 (28) |
| RPA | 1 (14) | 1 (14) | |
| Initial clinical data | Systolic BP (mmHg) | 121 [118–126] | 118 [116–121] |
| Diastolic BP (mmHg) | 64 [61.5–71] | 65 [61–71] | |
| Initial sFlt-1/PlGF ratio | 28 [16–47] | 21 [20–23] | |
| Aspirin treatment | 4 (57) | 3 (42) | |
| Initial obstetrics | Gravidity | 3 [2–4] | 3 [3–5] |
| Primigravida | 1 (14) | 0 (0) | |
| Parity | 1 [1–2] | 2 [1–2] | |
| GA at recruitment (weeks) | 12 [10–14] | 13 [12–13] | |
| Initial dietary data | Initial I-MEDAS score | 10 [9.5–12] | 10 [8–11] |
| Initial vitamin D (ng/mL) | 12 [9–21] | 22 [21–23] |
Continuous values are presented as median [IQR] and categorical values are presented as n (%). AID, autoimmune disease; BMI, body mass index; BP, blood pressure; CKD, chronic kidney disease; EVOOHEH, early harvested extra virgin olive oil; GA, gestational age; I-MEDAS, Mediterranean Diet Adherence Screener adopted to Israel; IQR, interquartile range; IVF, in vitro fertilization; RPA, regular physical activity; sFlt-1/PlGF, soluble fms-like tyrosine kinase 1 to placental growth factor; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Discussion
This interventional pilot study is designed to explore a novel dietary approach to mitigate the risk of PE in high-risk pregnant women through the daily addition of EVOOEH. Given the substantial burden of PE on maternal and perinatal health (1,4,5), and the current limitations of effective preventive strategies, investigating readily available and well-tolerated dietary interventions like EVOOEH is of paramount importance. Publishing this protocol at early recruitment stages can prompt early insights into the feasibility and acceptability of EVOOEH supplementation in high-risk pregnant women, including recruitment rates, adherence patterns (HT, I-MEDAS, RD calls), and safety/tolerability signals that are essential for optimizing protocol design.
PE believed to be progressed as a two-stage disorder integrating abnormal placentation with systemic maternal response (35). In the first stage, inadequate spiral artery remodeling leads to placental ischemia and heightened oxidative stress driven by excess ROS, which disrupts local vascular homeostasis (23,35). This milieu shifts the angiogenic balance toward reduced PlGF and VEGF and excess anti-angiogenic mediators such as sFlt-1 and soluble endoglin (sEng), promoting a profoundly anti-angiogenic state (21,35). In the second stage, these circulating factors induce widespread endothelial dysfunction, driving vasoconstriction, increased vascular permeability, and a prothrombotic tendency (8,21,22). Clinically, this translates into hypertension, proteinuria, and coagulopathy, accompanied by neurological and hepatic involvement that reflects the multisystem nature and severity of PE (35). We therefore propose EVOOEH intervention, due to its high antioxidant activity.
The rationale for using EVOOEH is firmly rooted in its rich antioxidant profile and its established anti-inflammatory and angiogenic modulating properties (18-20), which directly address key pathophysiological mechanisms underlying PE, namely oxidative stress, microvascular damage, and dysglycemia. By focusing on an early intervention (8 to 16 weeks gestation), we aim to influence placental development and maternal metabolic adaptations during a critical window, potentially preventing the cascade of events that lead to PE (21). The selection of study outcomes, including SBP, DBP, GCT results and sFlt-1/PlGF ratio, can directly reflects these underlying mechanisms and provides measurable markers of intervention efficacy.
A possible strength of this protocol is the use of HT as a compliance marker, which offers an objective measure of EVOO intake beyond self-report (29), enhancing the reliability of the intervention’s impact. The comprehensive collection of independent variables, including demographic, gynecological, obstetric, dietetic, and anthropometric data, will allow for robust multivariate analyses, accounting for potential confounders and providing a more nuanced understanding of the intervention’s effects. Furthermore, the single-masked design for data analysis minimizes potential bias.
This pilot study, while adequately powered for its diverse outcomes based on conservative estimates of PE incidence reduction, serves as a crucial preliminary step. Its findings can provide essential data regarding the feasibility, adherence, and initial efficacy of EVOOEH intervention. Positive results would strongly support the conduct of a larger, multi-center randomized controlled trial to confirm these findings and establish EVOOEH as a viable adjunctive strategy for PE prevention. Conversely, if no significant effects are observed, the study will still contribute valuable knowledge by ruling out this specific intervention under the tested conditions and inform future research directions in dietary prevention of PE. The integration of clinical data with advanced metabolomic analysis for olive oil phenols also represents a strength, bridging the gap between dietary intake and physiological response.
Nevertheless, several challenges must be acknowledged. First, ensuring consistent daily intake of EVOOEH throughout pregnancy, despite potential gastrointestinal sensitivities or changing food preferences, poses a real-world adherence concern. To mitigate this, the study includes regular dietary counseling, reminders, and the use of HT as a biochemical compliance marker, which together strengthen intervention fidelity. Second, the inherent variability in individual metabolic responses to polyphenol-rich foods, might obscure clear associations. The incorporation of advanced metabolomic profiling is a strategic solution, enabling a deeper exploration of biological responses beyond traditional biomarkers. Third, aspirin was co-administered to selected participants alongside interventions across both groups (Table 2), when indicated. This may pose a substantial confounding to the intervention, including EVOOHEH, as it can also lower PE risk (36). Hence, study outcomes, including those related to the intervention, should be interpreted with caution, considering the aforementioned challenges and the fact that these issues have only been partially addressed. Limitations inherent to pilot studies must also be recognized. The relatively small sample size, while appropriate for initial hypothesis generation and feasibility assessment, may limit the detection of more modest effects. Additionally, as a single-center study, generalizability could be constrained. Nonetheless, these limitations are offset by the study’s robust design elements, objective compliance measures, single-masked analysis, and integration of metabolomics, which together enhance data quality and interpretability.
In summary, this pilot study is strategically positioned to provide critical evidence on the feasibility and initial efficacy of EVOOEH in reducing risk factors for PE. Its results will directly inform the design of a future large-scale, multi-center randomized control trial (if ethically allowed), advancing the exploration of dietary strategies as accessible, well-tolerated interventions for improving pregnancy outcomes.
This pilot study protocol on the effect of EVOOEH in women at higher risk for PE can represent a significant step towards identifying focused and efficient dietary treatments for PE prevention. By evaluating its impact on PE incidence, GDM, and relevant biochemical markers, we anticipate generating crucial evidence that will inform future, larger-scale clinical trials and contribute to evidence-based nutritional guidelines for improving maternal and fetal outcomes in high-risk pregnancies.
Acknowledgments
EVOOHEH was supplied by the Olive Division at the Plant Council (Israeli Ministry of Agriculture). The authors wish to thank Dr. Arie Budovsky from BUMCA’s Research and Development Authority for fruitful discussions regarding this protocol. We extend our gratitude to Mr. Moatasem Ballh Zwahra from BUMCA’s Laboratory of Clinical Biochemistry and Mrs. Gali Menashe from the SEED Laboratory, Department of Nutrition Sciences, Faculty of Health Sciences, Ariel University for their assistance with bio-sampling administrative procedures and data collection. We also thank Ruhama Kremer and Hagit Afuta of BUMCA’s Department of Obstetrics and Gynecology for their assistance with the study’s ethical and administrative procedures.
Footnote
Reporting Checklist: The authors have completed the SPIRIT reporting checklist. Available at https://atm.amegroups.com/article/view/10.21037/atm-25-122/rc
Peer Review File: Available at https://atm.amegroups.com/article/view/10.21037/atm-25-122/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://atm.amegroups.com/article/view/10.21037/atm-25-122/coif). Y.S.O. initiated the study and has authority and control over the study (by the aid of BUMCA’s intramural fund). The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study will be conducted in accordance with the Declaration of Helsinki and its subsequent amendments. This study protocol has been approved by the Helsinki Committee of Barzilai University Medical Center at Ashkelon (BUMCA) (approval No. 0049-24-BRZ). All participants will provide informed written consent before enrollment.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Khan KS, Wojdyla D, Say L, et al. WHO analysis of causes of maternal death: a systematic review. Lancet 2006;367:1066-74. [Crossref] [PubMed]
- Magee LA, Nicolaides KH, von Dadelszen P. Preeclampsia. N Engl J Med 2022;386:1817-32. [Crossref] [PubMed]
- Wheeler SM, Myers SO, Swamy GK, et al. Estimated Prevalence of Risk Factors for Preeclampsia Among Individuals Giving Birth in the US in 2019. JAMA Netw Open 2022;5:e2142343. [Crossref] [PubMed]
- Burton GJ, Redman CW, Roberts JM, Moffett A. Pre-eclampsia: pathophysiology and clinical implications. BMJ 2019;366:l2381. [Crossref] [PubMed]
- Steegers EA, von Dadelszen P, Duvekot JJ, et al. Pre-eclampsia. Lancet 2010;376:631-44. [Crossref] [PubMed]
- Aouache R, Biquard L, Vaiman D, et al. Oxidative Stress in Preeclampsia and Placental Diseases. Int J Mol Sci 2018;19:1496. [Crossref] [PubMed]
- Goulopoulou S, Davidge ST. Molecular mechanisms of maternal vascular dysfunction in preeclampsia. Trends Mol Med 2015;21:88-97. [Crossref] [PubMed]
- McCance DR, Holmes VA, Maresh MJ, et al. Vitamins C and E for prevention of pre-eclampsia in women with type 1 diabetes (DAPIT): a randomised placebo-controlled trial. Lancet 2010;376:259-66. [Crossref] [PubMed]
- De-Regil LM, Palacios C, Ansary A, et al. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev 2012;2:CD008873. [Crossref] [PubMed]
- Purswani JM, Gala P, Dwarkanath P, et al. The role of vitamin D in pre-eclampsia: a systematic review. BMC Pregnancy Childbirth 2017;17:231. [Crossref] [PubMed]
- Brown MA, Magee LA, Kenny LC, et al. Hypertensive Disorders of Pregnancy: ISSHP Classification, Diagnosis, and Management Recommendations for International Practice. Hypertension 2018;72:24-43. [Crossref] [PubMed]
- Bartsch E, Medcalf KE, Park AL, et al. Clinical risk factors for pre-eclampsia determined in early pregnancy: systematic review and meta-analysis of large cohort studies. BMJ 2016;353:i1753. [Crossref] [PubMed]
- Aziz F, Khan MF, Moiz A. Gestational diabetes mellitus, hypertension, and dyslipidemia as the risk factors of preeclampsia. Sci Rep 2024;14:6182. [Crossref] [PubMed]
- ACOG Practice Bulletin No. 190: Gestational Diabetes Mellitus. Obstet Gynecol 2018;131:e49-64. [Crossref] [PubMed]
- Simmons D, Immanuel J, Hague WM, et al. Treatment of Gestational Diabetes Mellitus Diagnosed Early in Pregnancy. N Engl J Med 2023;388:2132-44. [Crossref] [PubMed]
- Ma'ayeh M, Costantine MM. Prevention of preeclampsia. Semin Fetal Neonatal Med 2020;25:101123. [Crossref] [PubMed]
- US Preventive Services Task Force. Screening for Gestational Diabetes: US Preventive Services Task Force Recommendation Statement. JAMA 2021;326:531-8. [Crossref] [PubMed]
- Flynn MM, Tierney A, Itsiopoulos C. Is Extra Virgin Olive Oil the Critical Ingredient Driving the Health Benefits of a Mediterranean Diet? A Narrative Review. Nutrients 2023;15:2916. [Crossref] [PubMed]
- Trombetta D, Smeriglio A, Marcoccia D, et al. Analytical Evaluation and Antioxidant Properties of Some Secondary Metabolites in Northern Italian Mono- and Multi-Varietal Extra Virgin Olive Oils (EVOOs) from Early and Late Harvested Olives. Int J Mol Sci 2017;18:797. [Crossref] [PubMed]
- Huang S, Wang Q, Wang Y, et al. Physicochemical characterization and antioxidant activities of Chongqing virgin olive oil: effects of variety and ripening stage. Food Measure 2020;14:2010-20.
- Silvani Y, Lovita AND, Maharani A, et al. Extra Virgin Olive Oil Modulates Vasodilator Enzyme Level by Repairing Angiogenesis Function in Rat Model of Preeclampsia. J Family Reprod Health 2020;14:38-44.
- Milena E, Maurizio M. Exploring the Cardiovascular Benefits of Extra Virgin Olive Oil: Insights into Mechanisms and Therapeutic Potential. Biomolecules 2025;15:284. [Crossref] [PubMed]
- Zhang L, Liu J, Feng X, et al. Unraveling the mysteries of spiral artery remodeling. Placenta 2023;141:51-6. [Crossref] [PubMed]
- Assaf-Balut C, García de la Torre N, Durán A, et al. A Mediterranean diet with additional extra virgin olive oil and pistachios reduces the incidence of gestational diabetes mellitus (GDM): A randomized controlled trial: The St. Carlos GDM prevention study. PLoS One 2017;12:e0185873. [Crossref] [PubMed]
- Gomez Ribot D, Diaz E, Fazio MV, et al. An extra virgin olive oil-enriched diet improves maternal, placental, and cord blood parameters in GDM pregnancies. Diabetes Metab Res Rev 2020;36:e3349. [Crossref] [PubMed]
- Abu-Saad K, Endevelt R, Goldsmith R, et al. Adaptation and predictive utility of a Mediterranean diet screener score. Clin Nutr 2019;38:2928-35. [Crossref] [PubMed]
- ACOG Committee Opinion No. 743: Low-Dose Aspirin Use During Pregnancy. Obstet Gynecol 2018;132:e44-52. [Crossref] [PubMed]
- Tsuprykov O, Buse C, Skoblo R, et al. Reference intervals for measured and calculated free 25-hydroxyvitamin D in normal pregnancy. J Steroid Biochem Mol Biol 2018;181:80-7. [Crossref] [PubMed]
- Pastor A, Rodríguez-Morató J, Olesti E, et al. Analysis of free hydroxytyrosol in human plasma following the administration of olive oil. J Chromatogr A 2016;1437:183-90. [Crossref] [PubMed]
- Fernandes S, Ribeiro C, Paiva-Martins F, et al. Protective effect of olive oil polyphenol phase II sulfate conjugates on erythrocyte oxidative-induced hemolysis. Food Funct 2020;11:8670-9. [Crossref] [PubMed]
- Wang R, Xu Q, Wang X, et al. Optimization of a preeclampsia early warning model driven by psychosocial factors: a five-year cohort study and its clinical impact. J Matern Fetal Neonatal Med 2025;38:2508903. [Crossref] [PubMed]
- Wainstock T, Sheiner E. Clinical factors associated with preeclampsia recurrence. Pregnancy Hypertens 2022;30:31-5. [Crossref] [PubMed]
- Available online: https://my.health.gov.il/CliniTrials/Pages/MOH_2024-11-27_013765.aspx, accessed on 28/06/2025
- An Interventional Pilot Study on the Effect of Extra Virgin Olive Oil on Women with Preeclampsia Risk. Available online: https://clinicaltrials.gov/study/NCT06759545, accessed on 28/06/2025
- Martini C, Saeed Z, Simeone P, et al. Preeclampsia: Insights into pathophysiological mechanisms and preventive strategies. Am J Prev Cardiol 2025;23:101054. [Crossref] [PubMed]
- Kupka E, Roberts JM, Mahdy ZA, et al. Aspirin for preeclampsia prevention in low- and middle-income countries: mind the gaps. AJOG Glob Rep 2024;4:100352. [Crossref] [PubMed]



