
Beyond the predictors of lymph node metastases in patients undergoing lymph node dissection for renal cell carcinoma: the impact of tumour side and location
The latest European Association of Urology (EAU) Guidelines on renal cell carcinoma (RCC) recommend offering extended lymph node dissection (LND) to patients with adverse clinical features and performing LND for staging purposes or local control in those with clinically enlarged lymph nodes (1). Of note, the strength of both recommendations is weak. Similarly, the expert opinion of the American Urological Association (AUA) renal cancer Guideline panel is that physicians should perform a lymph node dissection for staging purposes for patients with clinically concerning regional lymphadenopathy but not routinely in patients with localized RCC and clinically negative nodes (2).
These conflicting recommendations reflect the current controversial evidence on the indications, extent and prognostic impact of LND for RCC (3-6). Furthermore, they might partly explain the trend towards lower rates of LND over the last decades, with LND rates stabilizing after 2010 in both Europe and US (7,8).
Nowadays, key grey areas include: (I) the potential association of LND with survival in M0 and M1 patients; (II) the impact of LND template with survival outcomes; (III) the prognostic value of pN+ status; (IV) the association of LND with surgical morbidity; and (V) the (preoperative) prediction of patients most likely to harbor lymph node metastases. The most recent systematic review and meta-analysis addressing these topics found low-to-moderate quality evidence showing an independent prognostic role but no therapeutic benefit of LND in most M0 or M1 patients with RCC (3). Furthermore, available studies exploring the potential association of anatomic extent of LND with survival reached inconsistent results. Of note, a subset of high-risk nonmetastatic patients did experience long-term survival after surgical resection (3). In addition, in a recent study performance of LND at the time of nephrectomy improved patient’s risk stratification, resulting in a small, albeit nonnegligible, clinical benefit in selecting high-risk patients for further postoperative treatment (9).
While acknowledging the current controversies on LND in RCC, decision-making is often challenging in daily surgical practice and often relies on the surgeon’s individual clinical expertise. Indeed, as urologic surgeons, we ultimately have to balance the potential benefits and harms of LND in the individual patient. In this regard, while LND is not associated with an increased risk of perioperative morbidity in experienced hands (3,5,10), preoperative identification of patients with nodal metastases is a key unmet need.
This concept is reinforced by the finding that approximately 70% of patients showing suspected lymph nodes preoperatively do not harbor lymph node metastases at the final pathological analysis (11).
Several nomograms, risk scores and multivariable regression models have been proposed to identify patients at higher risk of lymph node invasion (LNI) (3,6). Although these tools are still suboptimal to guide decision-making, interestingly the most common covariates associated with pN+ disease are tumour size, stage and grade along with radiographic lymphadenopathy (cN1) (3).
In this scenario, Nini et al. should be commended for providing additional evidence to better contextualize the predictors of nodal metastases at surgery and/or nodal progression (NP) at follow-up in patients with RCC (12).
The study, which included 2,485 patients with sporadic, unilateral RCC treated with partial or radical nephrectomy at a single tertiary referral Centre with a median follow-up of 5 years, evaluated for the first time the potential association between tumour side and tumour location with the risk of LNI and/or NP.
The decision to perform LND was driven by the presence of ≥T2 and/or cN1 disease, as well as palpable lymph nodes during surgery, according to surgeon’s preference.
Regional LND included, on the right side, the renal hilar, pre- and retro-caval nodes, while on the left side, the renal hilar and para-aortic nodes. When an extended LND was sought, the inter-aorto-caval nodes were dissected beyond the regional template.
Overall, 372 (15%) and 279 (11%) patients had cN1 and cM1 disease, respectively. Notably, 43% of patients underwent LND with wither a regional (33%) or extended (10%) template. At final histopathological analysis, pN1 disease was reported in 7% of patients. The median number of lymph nodes removed was 6 (IQR, 3–10) in the overall cohort, while 5 and 12 in patients undergoing regional and extended LND, respectively (P<0.01).
The study provided novel data on the impact of tumour side and location on LNI and/or NP rates. In this regard, in the cohort of patients undergoing LND (n=1,061, 43%), LNI rates were 18% for right-sided RCC while 12% for left-sided RCC (P=0.02). If stratified by tumour location, LNI rates were 11%, 10%, 0%, 12% and 26% for upper, middle, hilar, lower area and more than one kidney area, respectively (P<0.01).
After stratification for both side and location, there was a statistically significant difference in LNI and/or NP rates among different tumour locations only when considering patients with tumours involving more than one area, which showed higher rates of nodal metastases as compared to all other single locations [Figures 1,2 of Ref. (12)].
In the subgroup analyses according to LND extent (no LND vs. regional vs. extended), patients with tumours involving multiple kidney areas not receiving LND had higher rates of NP as compared to patients with tumours involving single locations. On the contrary, there was no statistically significant difference in LNI and/or NP rates among different tumour locations when considering patients undergoing either regional or extended LND. Similarly, no significant differences in LNI and/or NP rates were found between right-sided versus left-sided tumours in the analyses stratified by LND extent.
The sub-analyses excluding patients with pT1a RCC confirmed these findings.
Importantly, at multivariable analyses, pathological tumour size, cM1, cN1, pT2 and pT3-T4 disease, as well as Fuhrman grade 3-4 were independent predictors of LNI and/or NP (all P≤0.01). Neither tumour side nor tumour location reached the independent predictor status.
The study by Nini et al. (12) provides key findings in the context of the current literature. First, the study shed light on the potential impact of tumour side and location on the risk of LNI and NP in patients undergoing surgery for RCC.
The rational for the study is grounded in the hypothesis that tumours with different sides and locations might have a different pattern of lymphatic drainage and consequently a different risk of LNI or NP. Intriguingly, despite patients with tumours involving multiple kidney locations appeared to have higher rates of nodal metastases in the analyses stratified by tumour side, tumor location was not associated with LNI or NP at final multivariable analysis (12). Moreover, as previously reported (1-3,6), the strongest predictors of nodal metastases at surgery were tumour size, stage and grade along with radiographic lymphadenopathy (cN1) and cM1 disease (3,11,13).
Taken together, these findings suggest that decision-making regarding indications and extent of LND for RCC should not rely on tumour side or location, but rather on selected “adverse clinical features” (1) which may aid surgeons in identifying patients most likely to harbor lymph node metastases at the time of nephrectomy.
Second, the study highlights the importance of reporting, beyond LNI at surgery, NP as a key endpoint during the follow-up of patients undergoing nephrectomy with or without LND for RCC. Yet, uniform definitions of NP according to standardized imaging criteria are lacking.
Third, the study highlights the unpredictable variability of lymphatic drainage from RCC (4,14). As previously reported for upper tract urothelial carcinoma (15), the pattern of lymphatic spread from RCC is highly heterogeneous (16). We recently found that, in light of this variability, the extent of the most commonly dissected templates might be insufficient to catch the anatomic pattern of lymphatic drainage from RCC, especially for higher stage and right-sided tumors (4). In this view, the study by Nini et al. (12) further reinforce the concept that, while different sides have different drainage patterns and require different LND templates (4), prediction of nodal status and the extent of dissection cannot ultimately rely on tumour location.
Despite the template for regional and extended LND reported by Nini and colleagues was codified and included the most critical anatomical sites (4,12), variability at a surgeon level might have impacted the number of nodes removed and the LNI and/or NP rates, potentially limiting the generalizability of study findings. Furthermore, as the probability of missing a positive node decreases with increasing number of LNs removed and the number of LNs needed for correct pN0 staging increases with higher pathological tumor stage and grade (17), the template reported by the Authors might have been still suboptimal to adequately assess LNI at surgery in some patients.
While waiting for more accurate models to preoperatively predict the risk of LNI (6), we believe the decision to perform LND should be carefully tailored according to the individual clinical scenario, taking into account patient (life expectancy and comorbidity status), tumour [adverse clinical features (1,3)] as well as surgeon (experience and skills) factors. We acknowledge that only a subset of high-risk patients is likely to benefit from LND (3), and that LND is a technically demanding procedure that might theoretically increase the risk of perioperative morbidity (5). Yet, LND is the most accurate staging tool in RCC, with the potential to improve risk-stratification and provide long-term cancer control in selected patients.
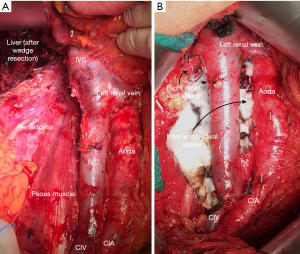
In light of the unpredictable nodal spread (4,5,16), we believe that, if performed, LND should always be extended and include the renal hilar, pre/para/retro-caval nodes (+/− inter-aortocaval nodes) for right-sided RCC while the renal hilar, pre/para-aortic and inter-aortocaval nodes for left-sided RCC, both from the crus of the diaphragm to the aortic bifurcation (Figure 1).
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Ljungberg B, Albiges L, Abu-Ghanem J, et al. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2019 Update. Eur Urol 2019;75:799-810. [Crossref] [PubMed]
- Campbell S, Uzzo RG, Allaf ME, et al. Renal Mass and Localized Renal Cancer: AUA Guideline. J Urol 2017;198:520-9. [Crossref] [PubMed]
- Bhindi B, Wallis CJD, Boorjian SA, et al. The role of lymph node dissection in the management of renal cell carcinoma: a systematic review and meta-analysis. BJU Int 2018;121:684-98. [Crossref] [PubMed]
- Campi R, Sessa F, Di Maida F, et al. Templates of Lymph Node Dissection for Renal Cell Carcinoma: A Systematic Review of the Literature. Front Surg 2018;5:76. [Crossref] [PubMed]
- Montgomery JS, Leibovich BC. Lymph node excision for renal cancer. J Urol 2013;189:419-21. [Crossref] [PubMed]
- Moschini M, Dell'Oglio P, Larcher A, et al. Lymph node dissection for renal carcinoma: what are we missing? Curr Opin Urol 2016;26:424-31. [Crossref] [PubMed]
- Capitanio U, Stewart GD, Larcher A, et al. European temporal trends in the use of lymph node dissection in patients with renal cancer. Eur J Surg Oncol 2017;43:2184-92. [Crossref] [PubMed]
- Farber NJ, Rivera-Núñez Z, Kim S, et al. Trends and outcomes of lymphadenectomy for nonmetastatic renal cell carcinoma: A propensity score-weighted analysis of the National Cancer Database. Urol Oncol 2019;37:26-32. [Crossref] [PubMed]
- Capogrosso P, Larcher A, Nini A, et al. The critical role of lymph node dissection in selecting high-risk nonmetastatic renal cancer candidates for adjuvant therapy after nephrectomy. Urol Oncol 2019;37:293.e25-293.e30. [Crossref] [PubMed]
- Gershman B, Moreira DM, Thompson RH, et al. Perioperative Morbidity of Lymph Node Dissection for Renal Cell Carcinoma: A Propensity Score-based Analysis. Eur Urol 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Capitanio U, Deho' F, Dell'Oglio P, et al. Lymphadenopathies in patients with renal cell carcinoma: clinical and pathological predictors of pathologically confirmed lymph node invasion. World J Urol 2016;34:1139-45. [Crossref] [PubMed]
- Nini A, Larcher A, Cazzaniga W, et al. The side and the location of the primary tumor does not affect the probability of lymph node invasion in patients with renal cell carcinoma. World J Urol 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Capitanio U, Abdollah F, Matloob R, et al. When to perform lymph node dissection in patients with renal cell carcinoma: a novel approach to the preoperative assessment of risk of lymph node invasion at surgery and of lymph node progression during follow-up. BJU Int 2013;112:E59-66. [Crossref] [PubMed]
- Kuusk T, De Bruijn R, Brouwer OR, et al. Lymphatic Drainage from Renal Tumors In Vivo: A Prospective Sentinel Node Study Using SPECT/CT Imaging. J Urol 2018;199:1426-32. [Crossref] [PubMed]
- Campi R, Minervini A, Mari A, et al. Anatomical templates of lymph node dissection for upper tract urothelial carcinoma: a systematic review of the literature. Expert Rev Anticancer Ther 2017;17:235-46. [Crossref] [PubMed]
- Karmali RJ, Suami H, Wood CG, et al. Lymphatic drainage in renal cell carcinoma: back to the basics. BJU Int 2014;114:806-17. [Crossref] [PubMed]
- Rieken M, Boorjian SA, Kluth LA, et al. Development and external validation of a pathological nodal staging score for patients with clear cell renal cell carcinoma. World J Urol 2018. [Epub ahead of print]. [Crossref] [PubMed]


