Predictive factors for immunotherapy in melanoma
Introduction
Skin cancer is one of the most common cancers worldwide (1,2). Melanoma accounts for less than 5% of all skin cancers but is the most aggressive form and responsible for the greatest number of skin cancer-related deaths at international level (3,4). Whereas most melanomas diagnosed at an early stage can often be treated with surgery alone, a proportion of these cancers may present a loco-regional or a systemic disease recurrence, conferring poor prognosis (5). The global incidence of melanoma is increasing worldwide, with a growing fraction of patients presenting with advanced disease. The clinical benefit from chemotherapy (for example, dacarbazine, temozolamide, fotemustine) in advanced melanoma patients is marginal, with a median duration of response of 7 months and an median overall survival (OS) under 1 year (6,7). The combination of different chemotherapeutic agents has not been demonstrated to affect patient survival, but only in terms of objective response rate (ORR) (8). However, in recent years, the landscape of melanoma treatment has radically changed with the introduction of targeted therapy and immunotherapy.
BRAF codon 600 mutations are detected in between 40% and 60% of melanoma patients, the majorities are the V600E mutation (80% of cases), V600K, V600R, etc. are also found. This genetic alteration represents a predictive biomarker of response to anti-BRAF (vemurafenib, dabrafenib) and anti-MEK kinase inhibitors (trametinib). Significant benefit in term of progression-free survival (PFS), OS and ORR in melanoma patients harboring BRAF mutations can be obtained with the use of these targeted agents (9). Almost invariably, however, the disease progresses after several months due to the emergence of acquired resistance (10-12). Melanoma patients negative for BRAF either have other mutations that are not good predictors of responses to specific inhibitors, such as NRAS or CKIT mutations (13,14), or do not harbor any actionable known molecular alteration treatable with targeted therapy.
Immunotherapy is a novel approach that is beginning to bear fruit and works by manipulating the patient’s endogenous immune system (often inhibited and repressed by the presence of a tumor) to react against cancer cells. Unlike anti-BRAF targeted therapy, the effectiveness of immune checkpoint inhibitors is not dependent on specific genetic alterations and may theoretically be applicable to all melanoma patients. However, only temporary and quantitative limited responses to immunotherapy agents have been demonstrated to date, making it a priority to identify those patients most likely to benefit (15,16). Here we review the current available literature regarding activity of immune checkpoint inhibitors in the treatment of advanced melanoma, with a focus on the potential predictive factors of response to anti-cytotoxic T-lymphocyte antigen-4 (CTLA-4) and programmed cell death-1 (PD-1) antibodies.
Evolution of immunotherapy in melanoma: FDA-approved agents
Based on the presence of anti-tumor immune cells within the tumor tissue, melanoma is considered to be a highly immunogenic disease due to the presence of anti-tumor immune cells within tumor tissue, which are a promising target for immunotherapy. One early milestone was the discovery of interleukin-2 (IL-2) in 1976. IL-2 is a cytokine produced by activated T cells that increases proliferation and activation of cytotoxic T cells, natural killer (NK) cells and monocytes. Immunotherapy with high doses of the immune molecule IL-2 induced long-term, durable, complete responses in a greater number of metastatic melanoma patients (20% of responses, with 5-7% complete responses) than had been previously achieved with dacarbazine, providing the first evidence of activity of immunotherapy in melanoma (17,18). However, to date, no prospective randomized phase III studies showing a survival benefit have been performed with IL-2. Nevertheless, in 1998 the US Food and Drug Administration (FDA) approved IL-2 for treatment of advanced melanoma. Another agent enhancing the immune system is interferon alpha (IFN-α), that showed a statistically significant improvement in both disease-free survival (DFS) and OS in adjuvant treatment of patients with high-risk cutaneous melanoma (19).
The second breakthrough in immune-based therapy was ipilimumab (Yervoy; Bristol-Myers Squibb, New York, US), a monoclonal antibody targeting CTLA-4 (20,21). In March 2011, the FDA approved this targeted agent for treatment of patients with newly diagnosed or previously treated unresectable or metastatic melanoma. This approval was based on a three-arm, multinational, randomized (3:1:1), double blind phase III clinical trial (NCT00094653) conducted in 676 patients with stage III/IV melanoma who experienced disease progression after standard treatment. A total of 403 patients were randomly assigned to ipilimumab plus a glycoprotein 100 (gp100) vaccine, ipilimumab alone, or gp100 alone. Results demonstrated that ipilimumab, with (10 months) or without (10.1 months) a gp100 peptide vaccine, improved OS in patients with previously treated metastatic melanoma compared with gp100 alone (6.4 months). However, only a fraction of patients achieve durable clinical responses that can last a decade and more (22).
Another targetable immune checkpoint is PD-1 and its ligand PD-L1. Antibodies targeting the PD-1/PD-L1 axis have shown promising clinical responses in melanoma. The most advanced antibodies against PD-1 receptor are nivolumab and pembrolizumab. A phase I clinical trial with nivolumab in 296 pretreated patients with solid tumors showed cumulative response rates of 28% among patients with metastatic melanoma (26 of 94) (23). Furthermore, a phase III double-blind trial with nivolumab showed significant improvements in PFS and OS in untreated BRAF wild type patients with advanced melanoma as compared with dacarbazine (24). The study showed a significant improvement in ORR (40% vs. 13.9%), PFS (5.1 vs. 22 months) and 1-year OS (72.95% vs. 42.1%) for the group of patients treated with nivolumab compared with those treated with dacarbazine. Nivolumab (Opdivo; Bristol-Myers Squibb) received FDA approval in December 2014 for patients with unresectable or metastatic melanoma and patients with disease progression following ipilimumab and, if patients are BRAF V600 mutation positive, a BRAF inhibitor.
A phase I clinical trial testing the activity of the PD-1 inhibitor pembrolizumab in patients with advanced melanoma, including those who progressed on ipilimumab, also showed a high rate of sustained tumor regression (25). In a further study in 173 patients with unresectable or metastatic melanoma with disease progression within 24 weeks of last dose of ipilimumab and, if BRAF V600 mutation positive, prior treatment with a BRAF inhibitor, it was demonstrated that patients treated with pembrolizumab experienced an ORR of 26% (26). These findings led to FDA approval of pembrolizumab (Keytruda; Merck Sharp & Dohme Corp, New Jersey, US) in September 2014 for treatment of patients with unresectable or metastatic melanoma who had progressed following ipilimumab and, for those with BRAF V600 mutation, following BRAF targeted therapy. To date, more than 100 clinical trials are ongoing testing immune-based therapies for melanoma and trying to determine which therapies can be optimally combined to achieve maximum possible efficacy.
Immune checkpoint therapy and available clinical data
Immune checkpoint therapy has led to important advances in cancer treatment. Rather than cancer cells, it targets molecules involved in regulation of T cells to maintain self-tolerance for removing inhibitory pathways used by tumors to escape immune surveillance. The immune system can both suppress tumor growth by eliminating cancer cells, but can also promote it by selecting for tumor cells able to evade surveillance (27). Cancer cells and tumor-infiltrating lymphocytes (TILs) resist immunoediting phase elimination by up-regulating the expression of inhibitory ligands and receptors that regulate T cell effector functions in a process known as T cell exhaustion (28). The inhibitory receptors on immune cells serve as immune checkpoints to prevent uncontrolled immune pathways. Immune checkpoints are initiated by ligand-receptor interactions and can be effectively blocked by monoclonal antibodies (mAbs), which have been shown to rescue otherwise exhausted antitumor T cells. The two most studied immune checkpoint receptors in cancer immunotherapy are CTLA-4, also known as CD152 and PD-1, also known as CD279; both inhibitory receptors regulate immune responses at different levels and by different mechanisms without overlapping and have distinct patterns of expression (29).
CTLA-4, a gene highly homologous to T cell costimulatory molecule CD28, is an important negative regulator of T cells. This receptor outcompetes CD28 for binding to B7 on antigen presenting cells (APC). Through interaction with its ligands CD80 (B7-1) and CD86 (B7-2), CD28 plays an important role in regulating amount of early activation of naive and memory T cells (30,31). On the other hand, PD-1 binds two B7 family ligands, known as PD-L1 (B7-H1) and PD-L2 (B7-DC) (32,33). In contrast to CTLA-4, interaction of PD-1 with its ligands functions to decrease the ability of already activated T cells in the periphery during the inflammatory response to infection in order to produce an effective immune response and prevent the immune system from rejecting the tumor (34).
Immune checkpoint inhibitors target either T cells or tumor cells to prevent attachment of each to the other so T cells stay activated. This allows activated T cells to infiltrate the tumor and attack cancer cells by their interaction and produce tumor responses, for example in patients with advanced melanoma. Currently, three immune checkpoint agents have been associated with objective clinical responses and have been approved by the FDA for treatment of melanoma (ipilimumab, nivolumab and pembrolizumab). Ipilimumab is the standard of care for patients with advanced melanoma and has achieved considerable improvement in OS for patients with metastatic melanoma when used as monotherapy in phase II and III trials (35,36) and in combination with other therapies (22,37) including cancer vaccines (38).
Another anti-CTLA-4 antibody is tremelimumab, which blocks binding of the APC ligands CD-80 and CD-86 to CTLA-4, allowing them to bind to another T cell surface receptor protein and induce T cell activation. Tremelimumab stimulates the immune system to destroy cancer cells. A phase I dose-escalation study indicated that tremelimumab can safely be administered at doses sufficient to generate antitumor responses in patients with advanced melanoma (39). Although phase I/II clinical studies have induced durable tumor responses in patients with metastatic melanoma (40,41), a phase III trial failed to demonstrate a statistically significant survival advantage in first-line treatment of patients with metastatic melanoma with single-agent tremelimumab over standard chemotherapy (temozolomide or dacarbazine) (42). Nonetheless, tremelimumab has been and is being studied in several clinical trials as single-agent or in combination with other agents in solid tumors. In a small size phase II trial, tremelimumab has shown clinical activity in advanced malignant mesothelioma and was recently approved (April 2015) by the FDA to treat malignant mesothelioma (43).
Multiple mAbs against PD-1 and its ligand (PD-L1) are currently in development and have shown great promise in melanoma and other malignancies. Considering historical data obtained—typical median OS of 6.2 months (95% CI, 5.9-6.5 months), 1-year OS of 25.5% (95% CI, 23.6-27.4%)—with ipilimumab for advanced melanoma (22), the results achieved with anti-PD-1 therapy represent a huge improvement in clinical benefit for these patients. Recently, two very important studies were published in this field. In the first, to identify the most effective immunotherapy treatment, investigators tested ipilimumab monotherapy against pembrolizumab or ipilimumab in combination with nivolumab in metastatic melanoma patients. This phase III study published by Robert et al. assigned 834 patients in a 1:1:1 ratio to pembrolizumab every 2 or 3 weeks, or ipilimumab every three weeks. Pembrolizumab prolonged PFS in both pembrolizumab groups (47.3% and 46.4%, respectively) versus ipilimumab (26.5%), and OS was significantly better with pembrolizumab cohorts than ipilimumab (74.1%, 68.4% and 58.2%, respectively). Response rate was also better with pembrolizumab (33.7%, 32.9% and 11.9%, respectively). Drug-associated grades 3 to 5 adverse effects were lower in the pembrolizumab groups than ipilimumab and occurred in 13.3%, 10.1%, and 19.9% of the groups, respectively (44).
The other study, published by Postow and colleagues, assigned untreated patients in a 2:1 ratio to receive ipilimumab combined with either nivolumab or placebo (monotherapy) until disease progression or unacceptable toxic effects. ORR was significantly greater with ipilimumab plus nivolumab (61%) than with ipilimumab monotherapy (11%), with complete responses in 22% of patients who received combined treatment, while patients who received monotherapy had no complete responses (45). Grades 3 to 4 drug-related adverse effects were reported in 54% of patients who received the combination therapy compared with 24% of patients who received ipilimumab monotherapy. ORR in the combination group was even higher than that reported previously by Wolchok and colleagues, which may be explained by the fact that the assigned patients in this trial were untreated (46). The fact that CTLA-4 and PD-1 regulate distinct inhibitory pathways and have non-overlapping mechanisms of action may be another reason for the higher efficacy in the combination cohort versus monotherapy. This was shown by Curran et al. in in vivo models: combination of CTLA-4 and PD-1 blockade was more than twice as effective as either CTLA-4 or PD-1 alone in driving tumor rejection of B16 melanomas models (47). Similar results for ORR were observed in 33 patients with BRAF-mutated tumors, consistent with the results of a previous phase I study (48). Both of these studies clearly show greater efficacy of anti-PD-1 mAbs for advanced melanoma.
Emerging issues—response evaluation (ircriteria), new antigenic agents
There is a high expectation that these immune checkpoint inhibitors, and others in this class, currently under investigation as melanoma immunotherapy, will also soon be approved for treatment of patients with melanoma and other tumor types (Table 1). Pathways that play a role in cancer immunotherapy are becoming increasingly well characterized; the use of new checkpoint inhibitors combinations, such as those combining anti-CTLA-4, anti-PD-1 or anti-LAG-3, including targeted therapies, are being tested to increase PFS and OS. All these options make immunotherapy look like an extremely promising for melanoma. To determine the expected benefit with immunotherapy in an unselected population, several predictive factors have been evaluated and/or are currently under investigation for efficacy and toxicity.

Full table
Predictive markers of anti-CTLA-4 therapy
There are currently no confirmed biomarkers predictive of response to anti-CTLA-4 studies. The hypothesis that HLA status could correlate with benefit from ipilimumab was refuted in a retrospective analysis of four advanced melanoma trials which found that ipilimumab’s mechanism of action was HLA independent and that similar OS was observed regardless of HLA subtype (49).
The debate currently rages as to whether the effector T cell or regulatory T cell (Treg) compartment is the primary target of CTLA-4 antibody-mediated blockade. Based on mouse melanoma models, investigators have found that concomitant blockade of both effector T cell and Tregs compartments contributes to the antitumor activity of CTLA-4 inhibitors, suggesting that ipilimumab’s primary antitumor effect may be targeting Tregs for destruction within the tumor microenvironment (50). In support of this finding, Hamid et al. found no associations between genetic polymorphisms and clinical activity, but did find significant associations between clinical efficacy and high baseline expression of FoxP3, indoleamine 2,3-dioxygenase (IDO) and increase in TILs between baseline and 3 weeks after starting ipilimumab treatment (51).
Whereas a single institution experience conducted in patients treated with ipilimumab demonstrated a significant correlation between survival and absolute lymphocyte count (52), others have shown that nearly all patients treated with ipilimumab had a significant increase in absolute lymphocyte count and that this occurrence was not specifically predictive of OS benefit from ipilimumab (53,54).
A preliminary study in melanoma showed that increase in myeloid-derived suppressor cell (MDSC) number in the peripheral blood by week 24 from pre-treatment baseline was associated with lack of clinical efficacy, suggesting that MDSC may serve both as a predictive and pharmacodynamic marker of treatment outcome (55). However, many IFN-γ-inducible genes and Th1-associated markers showed higher baseline expression in the tumor microenvironment of metastatic melanoma patients who benefited from ipilimumab treatment, suggesting that the genes of this particular type of T cells may serve as early predictors of response rather than predictive markers (56). Moreover, investigators have proposed that increased expression of the inducible co-stimulator (ICOS) molecule after anti-CTLA-4 therapy treatment can be used as a pharmacodynamic biomarker to assess adequate biologic response to treatment (monotherapy or combination) (57,58).
Last year, Snyder et al. published what appears to be the first reliable method to predict which melanoma patients will respond to anti-CTLA-4 therapy (ipilimumab and tremelimumab) (59). They discovered a neoantigen peptide landscape that is specifically present in tumors with a strong response to CTLA-4 blockade, and found an association between high mutational load and clinical benefit with CTLA-4 immune checkpoint inhibitors; however, this alone was not sufficient to predict outcome. This suggests that T cells can recognize as non-self (and therefore become more reactive to) tumors that express new antigens (neoepitopes) as a result of high number of non-driver genetic alterations (60,61).
Predictive biomarkers for the PD-1/PD-L1 pathway inhibitors
There is increasing evidence to support the hypothesis that an immune-active tumor microenvironment potentially correlates with improved melanoma patient survival or response to PD-1/PD-L1 pathway inhibitors. Selective infiltrations of CD8 positive T cells have been found to precede melanoma response to anti-PD1 therapy (62). Moreover, Tumeh et al. have demonstrated that pre-existing CD8 positive T cell density distinctly located at the invasive tumor margin in melanoma patients was associated with expression of the PD-1/PD-L1 immune inhibitory axis. This was found to be the best predictive parameter of clinical response to PD-1 blocking therapies followed by tumor CD8 positive density, whereas the poorest predictor was CD4 positive T cell density at the tumor and invasive margin (63).
PD-L1 expression is being investigated as a predictive biomarker of response for PD-1/PD-L1 directed therapy (Table 2). PD-L1 is expressed in several tumor types, including melanoma, lung, renal, kidney, head and neck and bladder cancer. Preliminary molecular marker studies in melanoma have shown a correlation of PD-L1 expression in pretreatment tumor specimens and objective response to anti-PD-1 therapy (23). However, PD-L1 expression in some studies appears to be associated with better prognosis only in metastatic melanoma lesions, suggesting that its predictive value may not be as clear-cut as initially thought (71). Other issues also add complexity when evaluating different analyses of PD-L1 expression as a predictive factor of response. PD-L1 expression is IFN-γ–inducible and can be present on either the tumor or infiltrating immune cells. Furthermore, there is currently no standardized methodology to measure PD-L1 expression and its evaluation differs between assays.
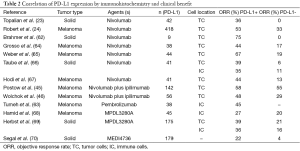
Full table
Taube and colleagues have found a significant correlation between the presence of TILs and PD-L1 expression in the tumor microenvironment. The number, type and location of TILs in primary tumors seem to have prognostic value and its presence may be more important for predicting response than PD-L1 expression alone. However, there is evidence that TILs are necessary but not sufficient for PD-L1 expression in melanoma (71). Patients with better response to these therapies are those that express high levels of PD-L1 and have infiltration of T cells within the tumor. Therefore, evaluation of PD-L1 expression by immunohistochemistry (IHC) together with measurement of immune infiltration might be a good predictor of tumor response to anti PD-L1 agents (72). However, there are caveats regarding measuring levels of PD-L1 since its expression is constitutive and its overexpression in response to stimuli can vary according to cell type. In addition, tumors are heterogeneous and the sample used for the assay may not be representative of the whole tumor. For instance, various levels of PD-L1 expression have been found in different metastases and their primary clear cell renal cell carcinomas (73). Moreover, it has been observed that patients with PD-L1 negative tumors can also respond to PD-1 and PD-L1 blockade (46,64,68). For all these reasons, a standardized definition of PD-L1 positivity that links these different assays is needed to evaluate PD-L1 expression as a predictive factor for PD-1 and/or PD-L1 pathway blockade.
Other immune biomarkers have also been assessed. Messina and colleagues found a direct correlation between a 12-chemokine gene expression signature and the presence of lymph nodal structures (immune cells that infiltrate and organize into intratumoral structures which resemble lymph nodes) associated with better OS in melanoma patients, something which may be useful in selecting those patients most suitable for immunotherapy (74).
Finally, tissue studies have demonstrated that tumors with a high somatic mutational frequency (above 10 somatic mutations per megabase of coding DNA), such as melanomas, respond best to PD-1 immune checkpoint inhibitors (75). As commented above, Snyder et al. have published similar results with CTLA-4 immunotherapy. The mutational load in melanoma has been found to be associated with clinical benefit but not predictive of response to treatment (59). These mutations may result in the presentation of neoantigens recognizable to the immune system and form a component of a predictive biomarker model of response to checkpoint blockade (76).
Conclusions
Recent advances in immunotherapy have revolutionized the spectrum of treatment options for melanoma patients along with surgery, radiotherapy, chemotherapy and targeted therapy. Immune checkpoint inhibitors are effective cancer treatments and have shown antitumor activity in several clinical studies with melanoma patients. However, not all patients benefit equally and efforts to identify predictive factors of clinical response are ongoing. Blocking the PD-1/PD-L1 pathway with monoclonal antibodies has shown better antitumor responses and safety profile (less toxicity) in clinical studies than has been seen with prior immunotherapies, such as IL-2 and anti-CTLA-4 agents. Nevertheless, long-term duration of benefit of either anti-PD-1 and/or PD-L1 agents is not as well-known as other treatments options such as the anti-CTLA-4 antibody ipilimumab which has been studied for longer and is better characterized. Evaluation of immunological biomarkers could offer useful prognostic information and facilitate clinical decision-making. It has been observed that characterizing tumors by immune infiltration (intratumoral infiltration), chemokine signature, tumor mutational load and PD-L1 expression, may be key molecular markers to assess the potential for selecting which patients may benefit from which immune checkpoint inhibitor, either in monotherapy or in combination, and may suggest the mechanism of an individual’s tumorigenesis. Optimal agents to combine with immune checkpoint mAbs might be those capable of inducing immune infiltration into the tumor microenvironment. Furthermore, PD-L1 expression by IHC is currently the strongest predictive marker of clinical benefit for immune checkpoint therapy but data presented so far do not demonstrate PD-L1 to be a reliable single predictive marker as the epidermal growth factor receptor (EGFR) is for lung cancer or human epidermal growth factor receptor 2 (HER2) for breast cancer. A standardization of PD-L1 IHC is required to explore the relationship between its expression and impact of this on prognosis of melanoma patients treated with PD-1 and/or PD-L1 mAbs. Given that infiltration of TILs is important to obtain an effective antitumor immune response, some categorization of immune infiltration together with PD-L1 expression by IHC or other immunologic assays might help to better predict of tumor response, although the fact that PD-L1 negative patients can also response means clinical application should be approached with caution. Finally, the identification and application of such possible predictive markers for each patient are crucial for the rational development, research and advance of immunotherapy and to guide the optimal choice of immunotherapy treatment.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin 2015;65:5-29. [PubMed]
- Chang AE, Karnell LH, Menck HR. The National Cancer Data Base report on cutaneous and noncutaneous melanoma: a summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 1998;83:1664-78. [PubMed]
- Little EG, Eide MJ. Update on the current state of melanoma incidence. Dermatol Clin 2012;30:355-61. [PubMed]
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. [PubMed]
- Beddingfield FC 3rd. The melanoma epidemic: res ipsa loquitur. Oncologist 2003;8:459-65. [PubMed]
- Patel PM, Suciu S, Mortier L, et al. Extended schedule, escalated dose temozolomide versus dacarbazine in stage IV melanoma: final results of a randomised phase III study (EORTC 18032). Eur J Cancer 2011;47:1476-83. [PubMed]
- Cui C, Tang B, Guo J. Chemotherapy, biochemotherapy and anti-VEGF therapy in metastatic mucosal melanoma. Chin Clin Oncol 2014;3:36. [PubMed]
- Chapman PB, Einhorn LH, Meyers ML, et al. Phase III multicenter randomized trial of the Dartmouth regimen versus dacarbazine in patients with metastatic melanoma. J Clin Oncol 1999;17:2745-51. [PubMed]
- McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol 2014;15:323-32. [PubMed]
- Flaherty KT, Puzanov I, Kim KB, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med 2010;363:809-19. [PubMed]
- Chapman PB, Hauschild A, Robert C, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med 2011;364:2507-16. [PubMed]
- Flaherty KT, Infante JR, Daud A, et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N Engl J Med 2012;367:1694-703. [PubMed]
- Nazarian R, Shi H, Wang Q, et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature 2010;468:973-7. [PubMed]
- Tsao H, Chin L, Garraway LA, et al. Melanoma: from mutations to medicine. Genes Dev 2012;26:1131-55. [PubMed]
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 2012;12:252-64. [PubMed]
- Kim T, Amaria RN, Spencer C, et al. Combining targeted therapy and immune checkpoint inhibitors in the treatment of metastatic melanoma. Cancer Biol Med 2014;11:237-46. [PubMed]
- Rosenberg SA, Lotze MT, Muul LM, et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. N Engl J Med 1985;313:1485-92. [PubMed]
- Rosenberg SA, Packard BS, Aebersold PM, et al. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N Engl J Med 1988;319:1676-80. [PubMed]
- Mocellin S, Pasquali S, Rossi CR, et al. Interferon alpha adjuvant therapy in patients with high-risk melanoma: a systematic review and meta-analysis. J Natl Cancer Inst 2010;102:493-501. [PubMed]
- O'Day SJ, Hamid O, Urba WJ. Targeting cytotoxic T-lymphocyte antigen-4 (CTLA-4): a novel strategy for the treatment of melanoma and other malignancies. Cancer 2007;110:2614-27. [PubMed]
- Fong L, Small EJ. Anti-cytotoxic T-lymphocyte antigen-4 antibody: the first in an emerging class of immunomodulatory antibodies for cancer treatment. J Clin Oncol 2008;26:5275-83. [PubMed]
- Hodi FS, O'Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 2010;363:711-23. [PubMed]
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 2012;366:2443-54. [PubMed]
- Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 2015;372:320-30. [PubMed]
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 2013;369:134-44. [PubMed]
- Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet 2014;384:1109-17. [PubMed]
- Anagnostou VK, Brahmer JR. Cancer immunotherapy: a future paradigm shift in the treatment of non-small cell lung cancer. Clin Cancer Res 2015;21:976-84. [PubMed]
- Wherry EJ. T cell exhaustion. Nat Immunol 2011;12:492-9. [PubMed]
- Topalian SL, Drake CG, Pardoll DM. Immune Checkpoint Blockade: A Common Denominator Approach to Cancer Therapy. Cancer Cell 2015;27:450-61. [PubMed]
- Walunas TL, Lenschow DJ, Bakker CY, et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity 1994;1:405-13. [PubMed]
- Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med 1995;182:459-65. [PubMed]
- Dong H, Zhu G, Tamada K, et al. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med 1999;5:1365-9. [PubMed]
- Latchman Y, Wood CR, Chernova T, et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol 2001;2:261-8. [PubMed]
- Intlekofer AM, Thompson CB. At the bench: preclinical rationale for CTLA-4 and PD-1 blockade as cancer immunotherapy. J Leukoc Biol 2013;94:25-39. [PubMed]
- Weber J, Thompson JA, Hamid O, et al. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin Cancer Res 2009;15:5591-8. [PubMed]
- Wolchok JD, Neyns B, Linette G, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol 2010;11:155-64. [PubMed]
- Maio M, Grob JJ, Aamdal S, et al. Five-year survival rates for treatment-naive patients with advanced melanoma who received ipilimumab plus dacarbazine in a phase III trial. J Clin Oncol 2015;33:1191-6. [PubMed]
- Attia P, Phan GQ, Maker AV, et al. Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J Clin Oncol 2005;23:6043-53. [PubMed]
- Ribas A, Camacho LH, Lopez-Berestein G, et al. Antitumor activity in melanoma and anti-self responses in a phase I trial with the anti-cytotoxic T lymphocyte-associated antigen 4 monoclonal antibody CP-675,206. J Clin Oncol 2005;23:8968-77. [PubMed]
- Camacho LH, Antonia S, Sosman J, et al. Phase I/II trial of tremelimumab in patients with metastatic melanoma. J Clin Oncol 2009;27:1075-81. [PubMed]
- Kirkwood JM, Lorigan P, Hersey P, et al. Phase II trial of tremelimumab (CP-675,206) in patients with advanced refractory or relapsed melanoma. Clin Cancer Res 2010;16:1042-8. [PubMed]
- Ribas A, Kefford R, Marshall MA, et al. Phase III randomized clinical trial comparing tremelimumab with standard-of-care chemotherapy in patients with advanced melanoma. J Clin Oncol 2013;31:616-22. [PubMed]
- Calabrò L, Morra A, Fonsatti E, et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: an open-label, single-arm, phase 2 trial. Lancet Oncol 2013;14:1104-11. [PubMed]
- Robert C, Schachter J, Long GV, et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N Engl J Med 2015;372:2521-32. [PubMed]
- Postow MA, Chesney J, Pavlick AC, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med 2015;372:2006-17. [PubMed]
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med 2013;369:122-33. [PubMed]
- Curran MA, Montalvo W, Yagita H, et al. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci U S A 2010;107:4275-80. [PubMed]
- Kluger H, Sznol M, Callahan M, et al. Survival, response duration, and activity by BRAF mutation (MT) status in a phase 1 trial of nivolumab (anti-PD-1, BMS-936558, ONO-4538) and ipilimumab (IPI) concurrent therapy in advanced melanoma (MEL). Ann Oncol 2014;25:iv374.
- Wolchok JD, Weber JS, Hamid O, et al. Ipilimumab efficacy and safety in patients with advanced melanoma: a retrospective analysis of HLA subtype from four trials. Cancer Immun 2010;10:9. [PubMed]
- Peggs KS, Quezada SA, Chambers CA, et al. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J Exp Med 2009;206:1717-25. [PubMed]
- Hamid O, Schmidt H, Nissan A, et al. A prospective phase II trial exploring the association between tumor microenvironment biomarkers and clinical activity of ipilimumab in advanced melanoma. J Transl Med 2011;9:204. [PubMed]
- Ku GY, Yuan J, Page DB, et al. Single-institution experience with ipilimumab in advanced melanoma patients in the compassionate use setting: lymphocyte count after 2 doses correlates with survival. Cancer 2010;116:1767-75. [PubMed]
- Hodi FS, Butler M, Oble DA, et al. Immunologic and clinical effects of antibody blockade of cytotoxic T lymphocyte-associated antigen 4 in previously vaccinated cancer patients. Proc Natl Acad Sci U S A 2008;105:3005-10. [PubMed]
- Postow MA, Chasalow SD, Yuan J, et al. Pharmacodynamic effect of ipilimumab on absolute lymphocyte count (ALC) and association with overall survival in patients with advanced melanoma. J Clin Oncol 2013;31:abstr 9052.
- Kitano S, Postow MA, Cortez C, et al. Myeloid-derived suppressor cell quantity prior to treatment with ipilimumab at 10mg/kg to predict for overall survival in patients with metastatic melanoma. J Clin Oncol 2012;30:abstr 2518.
- Ji RR, Chasalow SD, Wang L, et al. An immune-active tumor microenvironment favors clinical response to ipilimumab. Cancer Immunol Immunother 2012;61:1019-31. [PubMed]
- Vonderheide RH, LoRusso PM, Khalil M, et al. Tremelimumab in combination with exemestane in patients with advanced breast cancer and treatment-associated modulation of inducible costimulator expression on patient T cells. Clin Cancer Res 2010;16:3485-94. [PubMed]
- Ng Tang D, Shen Y, Sun J, et al. Increased frequency of ICOS+ CD4 T cells as a pharmacodynamic biomarker for anti-CTLA-4 therapy. Cancer Immunol Res 2013;1:229-34. [PubMed]
- Snyder A, Makarov V, Merghoub T, et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N Engl J Med 2014;371:2189-99. [PubMed]
- Segal NH, Parsons DW, Peggs KS, et al. Epitope landscape in breast and colorectal cancer. Cancer Res 2008;68:889-92. [PubMed]
- van Rooij N, van Buuren MM, Philips D, et al. Tumor exome analysis reveals neoantigen-specific T-cell reactivity in an ipilimumab-responsive melanoma. J Clin Oncol 2013;31:e439-42. [PubMed]
- Brahmer JR, Drake CG, Wollner I, et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol 2010;28:3167-75. [PubMed]
- Tumeh PC, Harview CL, Yearley JH, et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 2014;515:568-71. [PubMed]
- Grosso J, Horak CE, Inzunza D, et al. Association of tumor PD-L1 expression and immune biomarkers with clinical activity in patients (pts) with advanced solid tumors treated with nivolumab (anti-PD-1; BMS-936558; ONO-4538). J Clin Oncol 2013;31:abstr 3016.
- Weber JS, Kudchadkar RR, Yu B, et al. Safety, efficacy, and biomarkers of nivolumab with vaccine in ipilimumab-refractory or -naive melanoma. J Clin Oncol 2013;31:4311-8. [PubMed]
- Taube JM, Klein A, Brahmer JR, et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res 2014;20:5064-74. [PubMed]
- Hodi FS, Sznol M, Kluger HM, et al. Long-term survival of ipilimumab-naive patients (pts) with advanced melanoma (MEL) treated with nivolumab (anti-PD-1, BMS-936558, ONO-4538) in a phase I trial. J Clin Oncol 2014;32:abstr 9002.
- Hamid O, Sosman JA, Lawrence DP, et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with locally advanced or metastatic melanoma (mM). J Clin Oncol 2013;31:abstr 9010.
- Herbst RS, Soria JC, Kowanetz M, et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014;515:563-7. [PubMed]
- Segal NH, Antonia SJ, Brahmer JR, et al. Preliminary data from a multi-arm expansion study of MEDI4736, an anti-PD-L1 antibody. J Clin Oncol 2014;32:abstr 3002^.
- Taube JM, Anders RA, Young GD, et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med 2012;4:127ra37.
- Sznol M, Chen L. Antagonist antibodies to PD-1 and B7-H1 (PD-L1) in the treatment of advanced human cancer--response. Clin Cancer Res 2013;19:5542. [PubMed]
- Callea M, Genega EM, Gupta M, et al. PD-L1 expression in primary clear cell renal cell carcinomas (ccRCCs) and their metastases. J Clin Oncol 2014;32:abstr 467.
- Messina JL, Fenstermacher DA, Eschrich S, et al. 12-Chemokine gene signature identifies lymph node-like structures in melanoma: potential for patient selection for immunotherapy? Sci Rep 2012;2:765. [PubMed]
- Lawrence MS, Stojanov P, Polak P, et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 2013;499:214-8. [PubMed]
- Schumacher TN, Schreiber RD. Neoantigens in cancer immunotherapy. Science 2015;348:69-74. [PubMed]


